Incidence
Types of Anthracycline Cardiotoxicity
Diagnosis
Diagnosis is based on:
Risk Assessment
The risk of anthracycline-related CTRCD depends on baseline cardiovascular factors, cancer type, and prior or planned therapies.
A comprehensive baseline CV risk assessment is recommended (https://www.heartscore.org/en_GB/ ), as well as cardiotoxicity risk assessment (https://www.cancercalc.com/hfa-icos_cardio_oncology_risk_assessment.php) .
Anthracycline equivalent dosis calculator: https://www.cancercalc.com/anthracycline.php
Prevention of Anthracycline-Related CTRCD
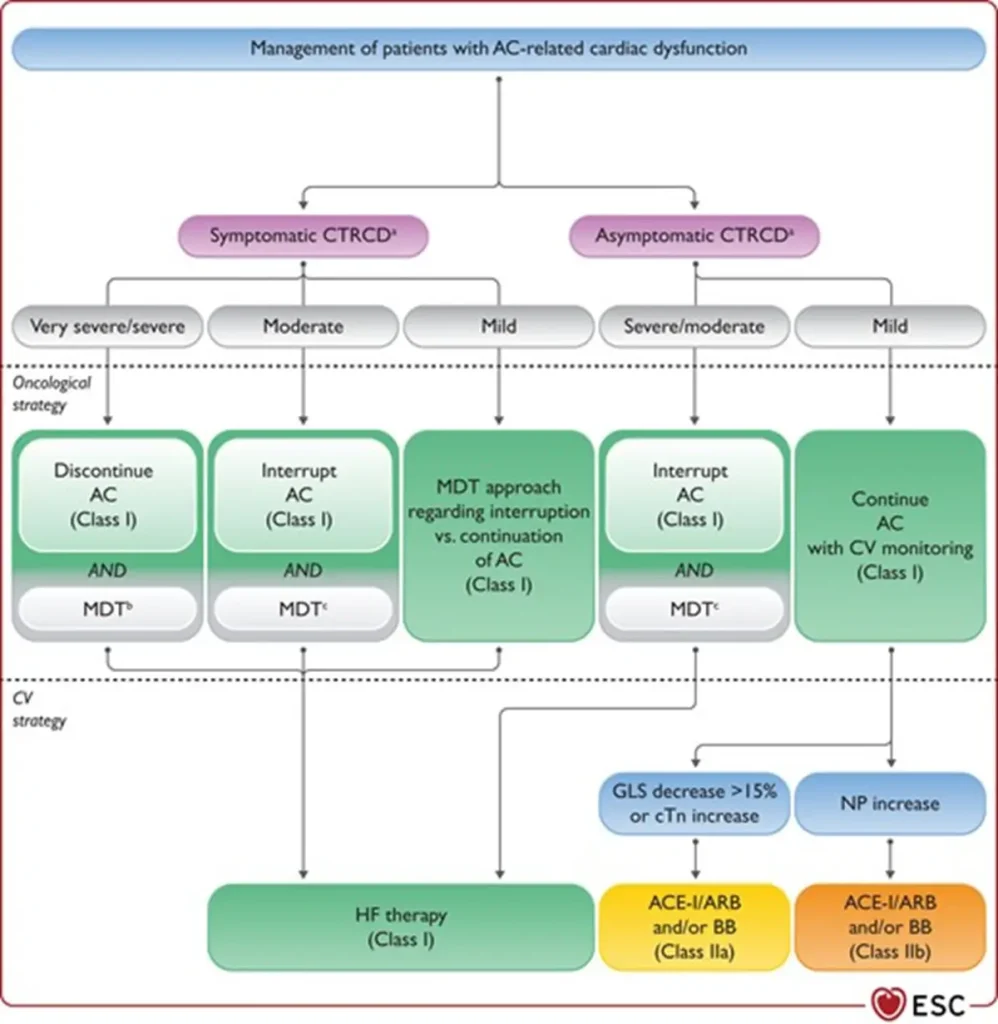
| Interruption of anthracycline therapy | ° Multidisciplinary discussion recommended to balance oncological benefit and cardiovascular risk after new CTRCD ° Discontinue anthracyclines in severe symptomatic CTRCD ° Temporarily interrupt anthracyclines in: – Moderate symptomatic CTRCD – Moderate or severe asymptomatic CTRCD |
| Heart failure therapy | Initiate and uptitrate guideline-directed HF therapy in: – Symptomatic moderate or severe CTRCD – Asymptomatic severe CTRCD |
| Mild asymptomatic CTRCD | Consider ACE-I, ARB, or beta-blocker while continuing anthracycline therapy uninterrupted |
| Exercise | Aerobic exercise is recommended before and during anthracycline therapy |
Permissive cardiotoxicity
Continuation or initiation of cardiotoxic cancer therapy (e.g. anthracyclines, trastuzumab) may be feasible in selected patients with mild–moderate cardiotoxicity or pre-existing cardiac disease.
Key elements of perimissive cardiotoxicity:
| Eligibility for anthracycline rechallenge | In patients with mild or moderate symptomatic CTRCD, or moderate/severe asymptomatic CTRCD, restarting anthracycline therapy after recovery of LV function should be evaluated in a multidisciplinary team (MDT) |
| Dose considerations | Consider dose reduction:
|
| Schedule modification | Consider:
|
| Formulation | Consider switching to liposomal anthracyclines |
| Cardioprotection | Consider dexrazoxane before each cycle (not reimbursed in Belgium) |
| Heart failure therapy | Aerobic exercise is recommended before and during anthracycline therapy |
| Cardiac monitoring | Close surveillance every 1–2 treatment cycles |
Permissive cardiotoxicity
Continuation or initiation of cardiotoxic cancer therapy (e.g. anthracyclines, trastuzumab) may be feasible in selected patients with mild–moderate cardiotoxicity or pre-existing cardiac disease.
Key elements of perimissive cardiotoxicity:
References
Incidence
Diagnosis
Diagnosis is based on:
Pathogenesis
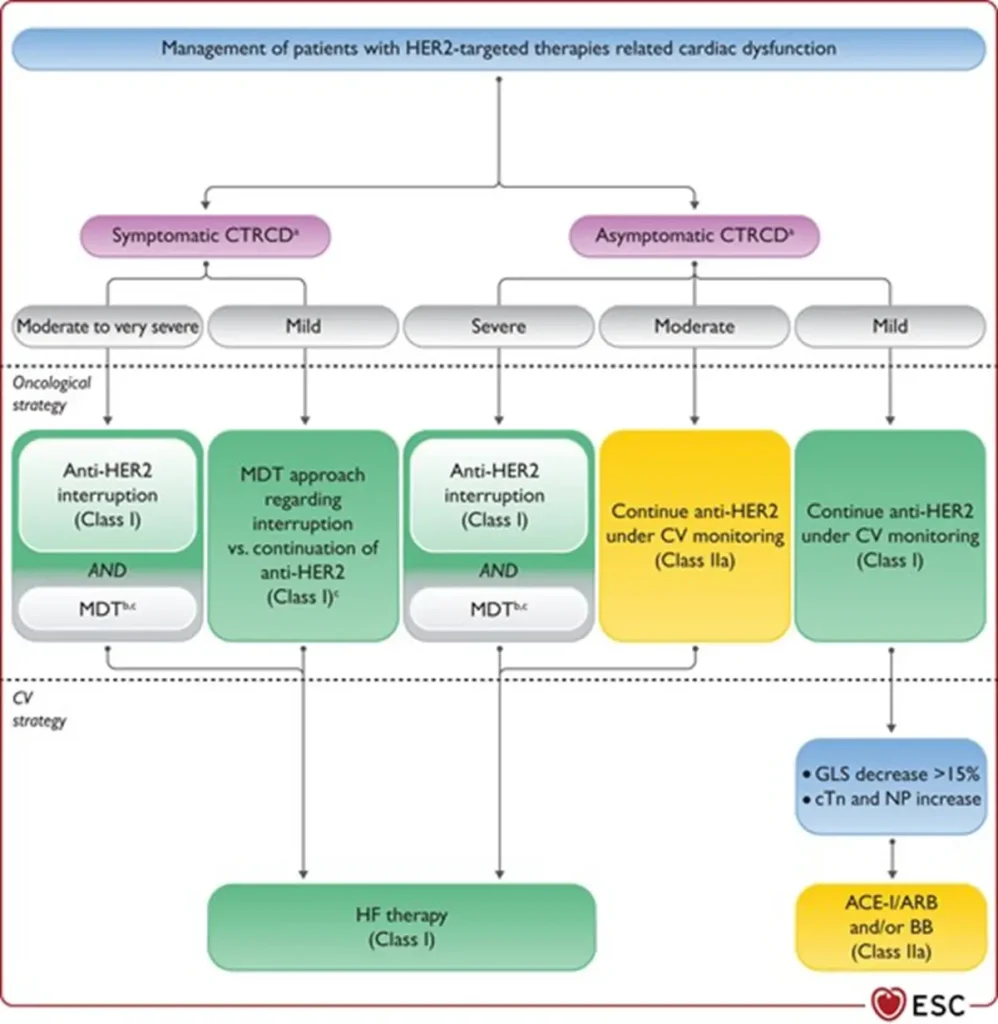
| Interruption of HER2-targeted therapy | Temporary interruption recommended for:
|
| Continuation of HER2 therapy | Asymptomatic moderate CTRCD (LVEF 40–49%):
Asymptomatic mild CTRCD (LVEF ≥50% with significant GLS decline and/or biomarker rise):
|
| Heart failure therapy | Initiate and uptitrate guideline-directed HF therapy in:
|
| Rechallenge after CTRCD recovery | Possible in patients continuing or restarting HER2-targeted therapy after recovery (LVEF ≥40%, ideally ≥50%):
|
Permissive cardiotoxicity
Continuation or initiation of cardiotoxic cancer therapy (e.g. anthracyclines, trastuzumab) may be feasible in selected patients with mild–moderate cardiotoxicity or pre-existing cardiac disease.
Key elements of perimissive cardiotoxicity:
implementation of cardioprotective strategies
References
| Drug Class | Agents |
| VEGF pathway inhibitors (VEGFi) | Bevacizumab, aflibercept, ramucirumab |
| Multitargeted TKIs with VEGF inhibition | Sunitinib, sorafenib, pazopanib, axitinib, lenvatinib, cabozantinib |
| BCR-ABL tyrosine kinase inhibitors | Imatinib, dasatinib, nilotinib, ponatinib, bosutinib |
| BRAF inhibitors / MEK inhibitors | Vemurafenib, dabrafenib / trametinib, cobimetinib |
| ALK inhibitors | Crizotinib, alectinib, brigatinib, lorlatinib |
| Proteasome inhibitors | Carfilzomib, bortezomib |
| Bruton tyrosine kinase (BTK) inhibitors | Ibrutinib |
| Fluoropyrimidines | 5-fluorouracil (5-FU), capecitabine |
| Platinum compounds | Cisplatin, carboplatin, oxaliplatin |
| Alkylating agents | Cyclophosphamide, ifosfamide |
| Aromatase inhibitors | Anastrozole, letrozole, exemestane |
| Androgen receptor pathway inhibitors (ARPI) | Abiraterone, bicalutamide, enzalutamide |
Treament
| Cancer Therapy | Avoid | Reason |
| ALL | verapamil, diltiazem | CYP3A4 inhibition |
| mTOR inhibotors | ACEi | angioedema |
| platinum-compounds | loop diuretics | oto- and nephrotoxicity |
| cyclophosphamide | thiazide | myelosuppression |
| abiraterone | spironolactone | paradoxical pro-androgenic (eplerenone is safe) |
References
Ventricular Arrhythmias (VA)
Overview
Ventricular arrhythmias are uncommon in cancer patients. The incidence increases with advanced disease and pre-existing cardiovascular comorbidities.
Proposed Mechanisms
Management of Cancer Therapy–Related VA
QTc Prolongation
Most cancer therapy–related VA are associated with QTc prolongation leading to torsade de pointes (TdP).
QTc prolonging cancer drugs and related risk factors are summarized in Tables 1 and 2
Normal QTc values
Clinical guidance
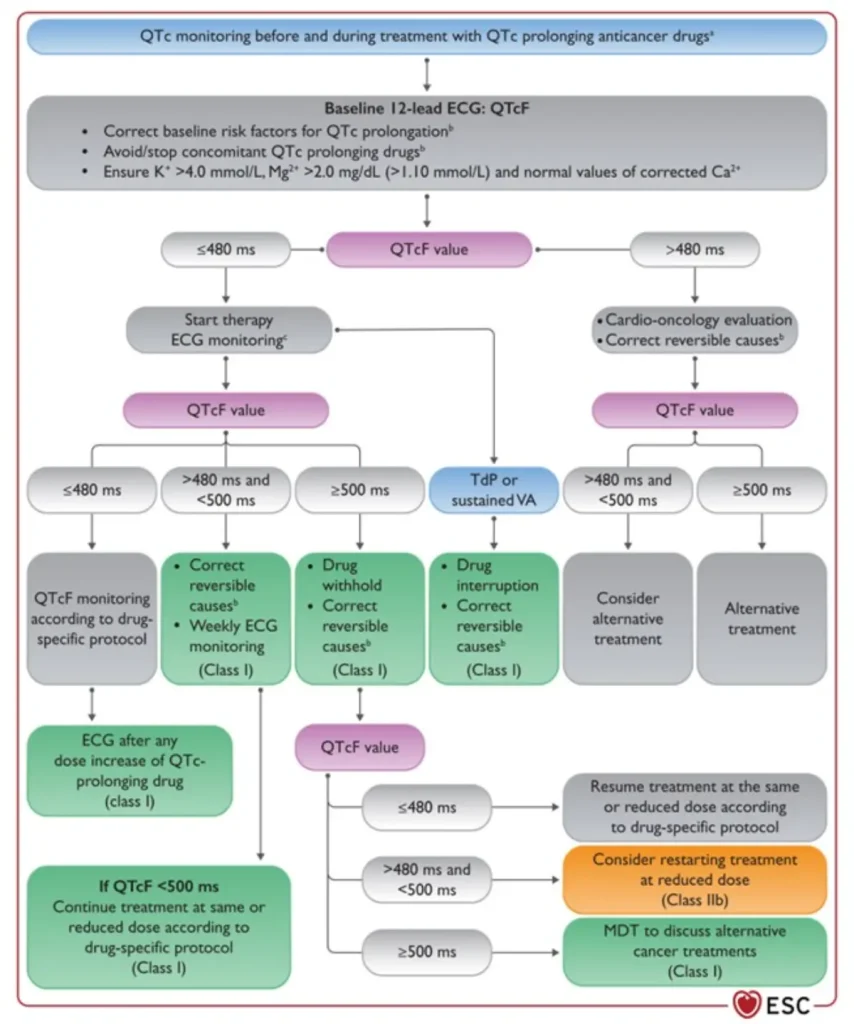
Rechallenge with QTc prolonging therapies
| Classification | Drugs |
| High risk (≥10 ms, TdP risk) | Aclarubicin, Arsenic trioxide, Glasdegib, Nilotinib, Oxaliplatin, Pazopanib, Ribociclib, Sunitinib, Toremifene, Vandetanib |
| Moderate risk (≥10 ms, low or uncertain TdP risk) | Abarelix, Belinostat, Brigatinib, Cabozantinib, Ceritinib, Crizotinib, Dovitinib, Entrectinib, Eribulin, Gilteritinib, Ivosidenib, Lapatinib, Lenvatinib, Osimertinib, Panobinostat, Rucaparib, Selpercatinib, Sorafenib, Tipiracil/Trifluridine, Vemurafenib |
| Low risk (<10 ms) | ADT, Afatinib, Axitinib, Binimetinib, Bortezomib, Bosutinib, Carfilzomib, Dabrafenib, Dasatinib, Encorafenib, Midostaurin, Pertuzumab, Ponatinib, Romidepsin, Quizartinib, Tamoxifen, Vorinostat |
| Correctable | Non-correctable |
QT-prolonging drugs | Acute myocardial ischaemia |
| Bradyarrhythmia | Age >65 years |
| Electrolyte abnormalities (↓K⁺ ≤3.5, ↓Mg²⁺ ≤1.6, ↓Ca²⁺ ≤8.5) | Baseline QTc prolongation |
| Inadequate dose adjustment for renal/hepatic clearance | Family history of sudden death (LQTS or genetic) |
| Female sex | |
| Impaired renal or hepatic function | |
| History of syncope or drug-induced TdP | |
| Pre-existing CVD (CAD, HF, LVH) |
References
Overview
Bradyarrhythmias may occur as a result of cancer therapy, most often with immune checkpoint inhibitors (ICI), immunomodulatory drugs (IMiD), and ALK inhibitors.
Mechanisms
Management
ICI-associated AV conduction disease
Drug-induced sinus bradycardia (IMiD, ALK inhibitors)
References
References
Commonly / frequently implicated therapies
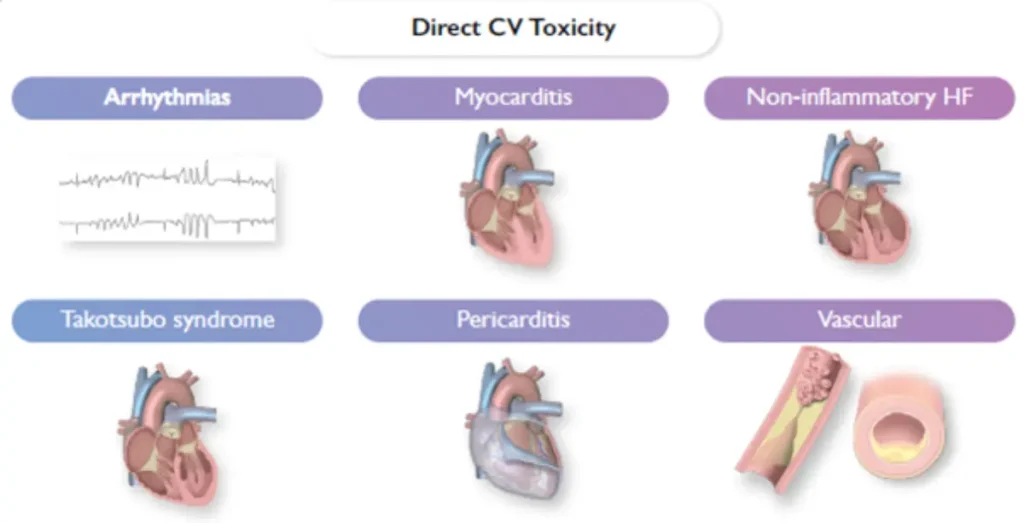
Most commonly encountered toxicities :
* : diagnosis and treatment according to current ESC guidelines + see specific topic.
# : most urgent diagnosis, specific therapeutic implications – see below.
ICI induced myocarditis:
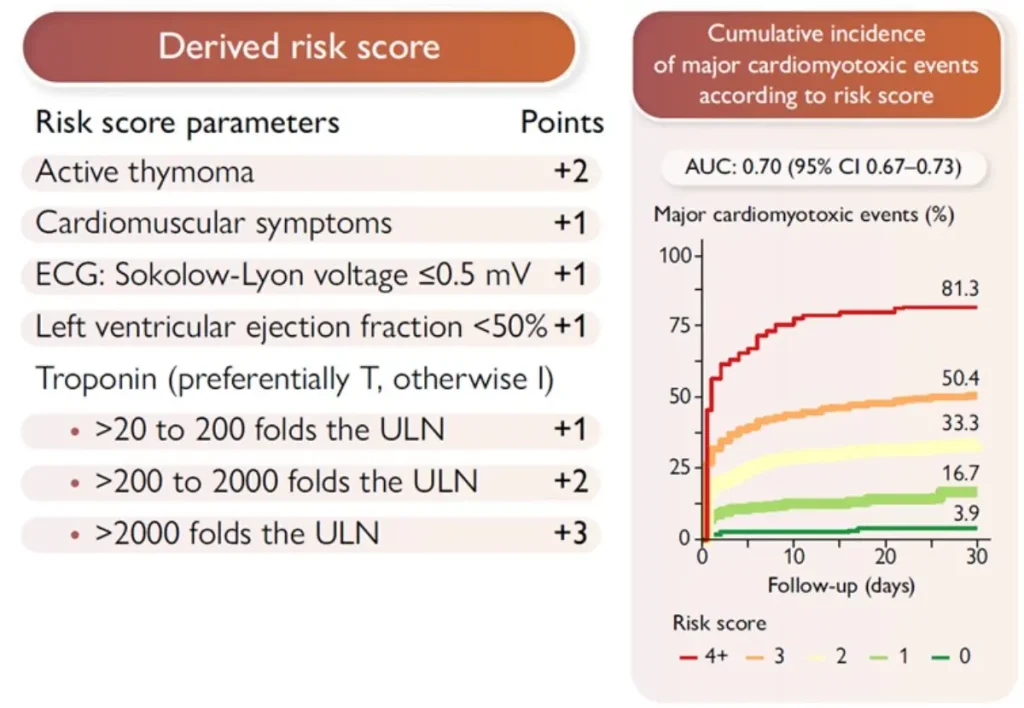
Diagnostic criteria for ICI-M : pathohistological diagnosis or clinical diagnosis :
| Required | Cardiac troponin (cTn) elevation (new or significant change from baseline) AND ≥1 major criterion OR ≥2 minor criteria, after exclusion of acute coronary syndrome (ACS) and acute infectious myocarditis based on clinical assessment. |
| Major criterion | Cardiac magnetic resonance (CMR) diagnostic for acute myocarditis according to modified Lake Louise criteria |
| Minor criteria |
|
Pathohistological diagnosis (endomyocardial biopsy) : Multifocal inflammatory cell infiltrates with overt cardiomyocyte loss by light microscopy
In order to exclude alternative or co-existing cardiac disease a hierarchical diagnostic approach in ICI myocarditis has been proposed:
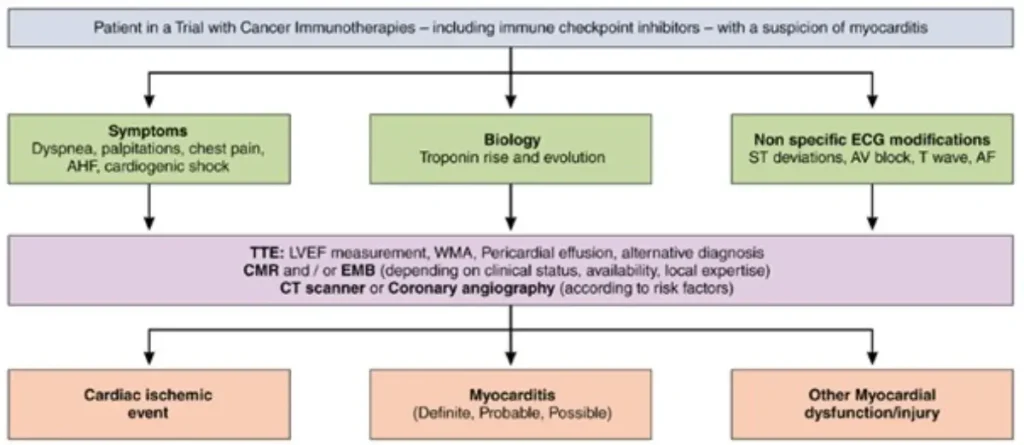

| Caveats | |
| Symptoms | Often non-specific;dyspnoea is the most common presenting symptom; chest pain may be absent |
| ECG | Always exclude ACS; ventricular arrhythmias or high ectopic burden and/or new conduction abnormalities (including AV block or PR prolongation) may indicate fulminant disease; continuous telemetry is recommended. |
| Transthoracic echocardiography (TTE | LVEF may be normal in ~50% of cases; GLS abnormalities are prognostic for MACE; pericardial effusion may be present but is not required for diagnosis |
| Cardiac biomarkers | Troponin I or T may be used; Troponin T may be falsely elevated with concomitant myositis; magnitude of troponin rise has prognostic value; NT-proBNP has limited diagnostic utility |
| Cardiac magnetic resonance (CMR) | May be falsely negative if performed early (<72 h after symptom onset); Repeat CMR if negative but clinical suspicion remains high |
| Endomyocardial biopsy (EMB) | Low threshold recommended in haemodynamic instability (cardiogenic shock / fulminant myocarditis) or when coronary angiography is performed |
| FDG-PET/CT | Conflicting evidence regarding diagnostic accuracy Repeat CMR if negative but clinical suspicion remains high |
| Severity category | Definition / Clinical features |
| Severe | Haemodynamic instability; heart failure requiring non-invasive or invasive ventilation; complete or high-grade atrioventricular block; and/or significant ventricular arrhythmias |
| Non-severe (clinically significant) | Symptomatic myocarditis with haemodynamic and electrical stability; may have reduced LVEF; no features of severe disease |
| Smouldering (subclinical) | Incidentally diagnosed myocarditis without clinical signs or symptoms |
| Steroid-refractory | Non-resolving or worsening myocarditis (clinical deterioration or persistent troponin elevation after exclusion of other causes) despite high-dose methylprednisolone |
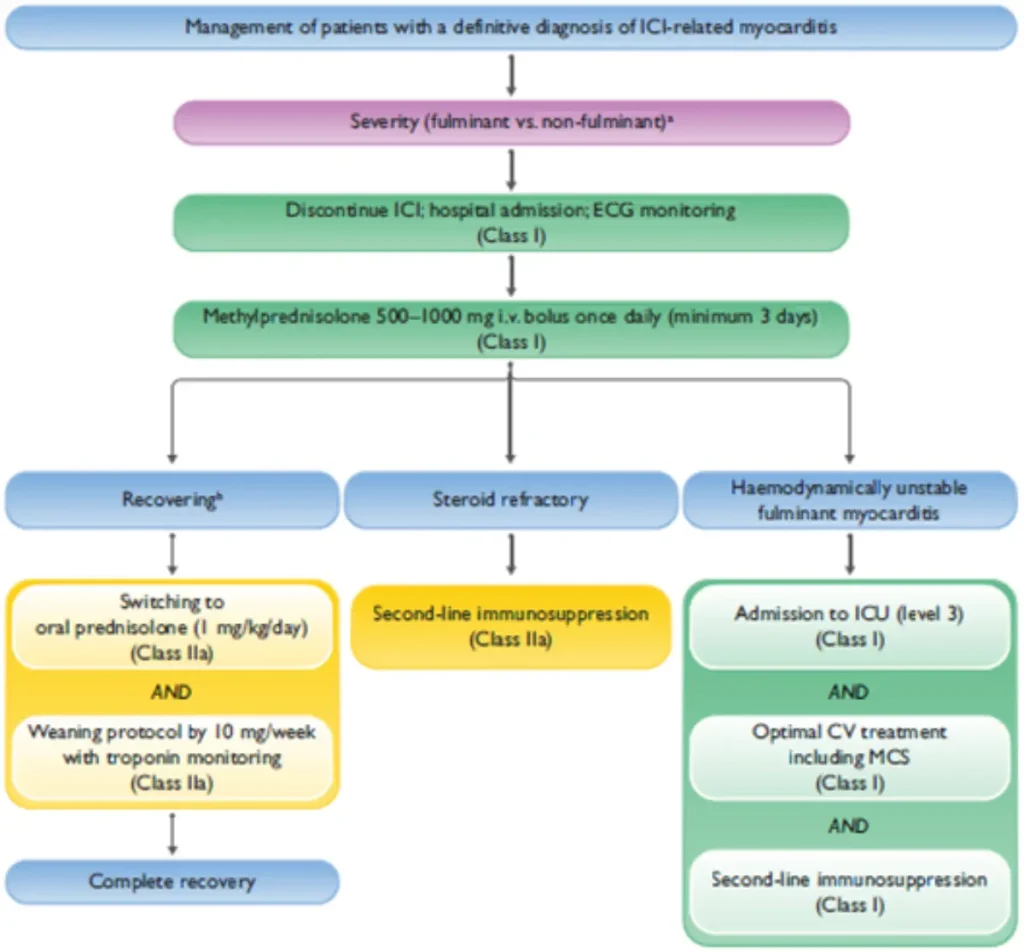
| Key points | |
| Steroid initiation | Do not delay corticosteroid therapy in case of diagnostic uncertainty; delayed treatment is associated with significantly worse prognosis |
| Cardiac monitoring |
|
| Immunosuppression escalation and second-line therapy options |
|
| Fulminant myocarditis / cardiogenic shock |
|
Oncological considerations |
|
Rechallenge : possibilities and caveats
ALWAYS discuss case with treating oncologist and/or BITOX :
In case of doubt :
Consider multidisciplinary discussion via BSMO BITOX Immunomanager submission
References
| Category | Key causes |
| Cancer-related | Direct invasion; metastases (lung, breast, melanoma, lymphoma/leukaemia, neighbouring organs); mediastinal lymphatic obstruction |
| Treatment-related | Chemotherapy (anthracyclines, cyclophosphamide, bleomycin, cytarabine); thoracic radiotherapy; targeted therapies (ATRA, arsenic trioxide, TKIs—especially dasatinib); immune therapies (IL-2, IFN-α, ICI); combination therapy ↑ risk |
| Primary pericardial malignancy | Rare; pericardial mesothelioma |
| Infectious | Mainly in immunocompromised patients |
| Other causes | Trauma, cardiac disease, renal failure, thyroid dysfunction (esp. hypothyroidism), idiopathic |
Symptoms
Symptoms are similar to the general population and range from typical pericarditic chest pain (worse when supine or with deep inspiration, relieved by sitting forward) to dyspnoea and fatigue with pericardial effusion, which may be asymptomatic. Cardiac tamponade is a medical emergency, presenting with severe dyspnoea, hypotension, tachycardia, syncope, and signs of poor peripheral perfusion
| Domain | Key points |
| ECG | ECG may show diffuse ST elevation and PR depression (pericarditis); low QRS voltages and electrical alternans (large effusion/tamponade); reactive atrial fibrillation may occur |
| Laboratory tests | CRP; high-sensitivity troponin I or T to exclude myocardial involvement; additional tests guided by differential diagnosis |
| Initial imaging | Chest X-ray may show enlarged cardiac silhouette |
| Cardiac imaging | Transthoracic echocardiography (gold standard) to assess effusion size and haemodynamic impact |
| Advanced imaging (CT / CMR) | Detailed assessment of pericardial inflammation; identification of local tumour invasion or masses |
| Diagnostic confirmation in effusion | Pericardial fluid cytology and/or pericardial biopsy required to confirm or exclude malignant involvement |
| Tumour markers | CEA, CYFRA 21-1, NSE, CA 19-9 may support diagnosis but lack diagnostic accuracy |
| Important note | ~Two-thirds of pericardial effusions in cancer patients are non-malignant → cytology remains essential |
Treatment
A multidisciplinary approach (cardio-oncology, oncology, radiotherapy, surgery) is recommended.
Acute pericarditis
ICI-related pericarditis
Rechallenge after resolution → MDT decision with close monitoring
Pericardial Effusion
General
Short term prognosis
Long-term risks
References
Association with cancer
Diagnosis
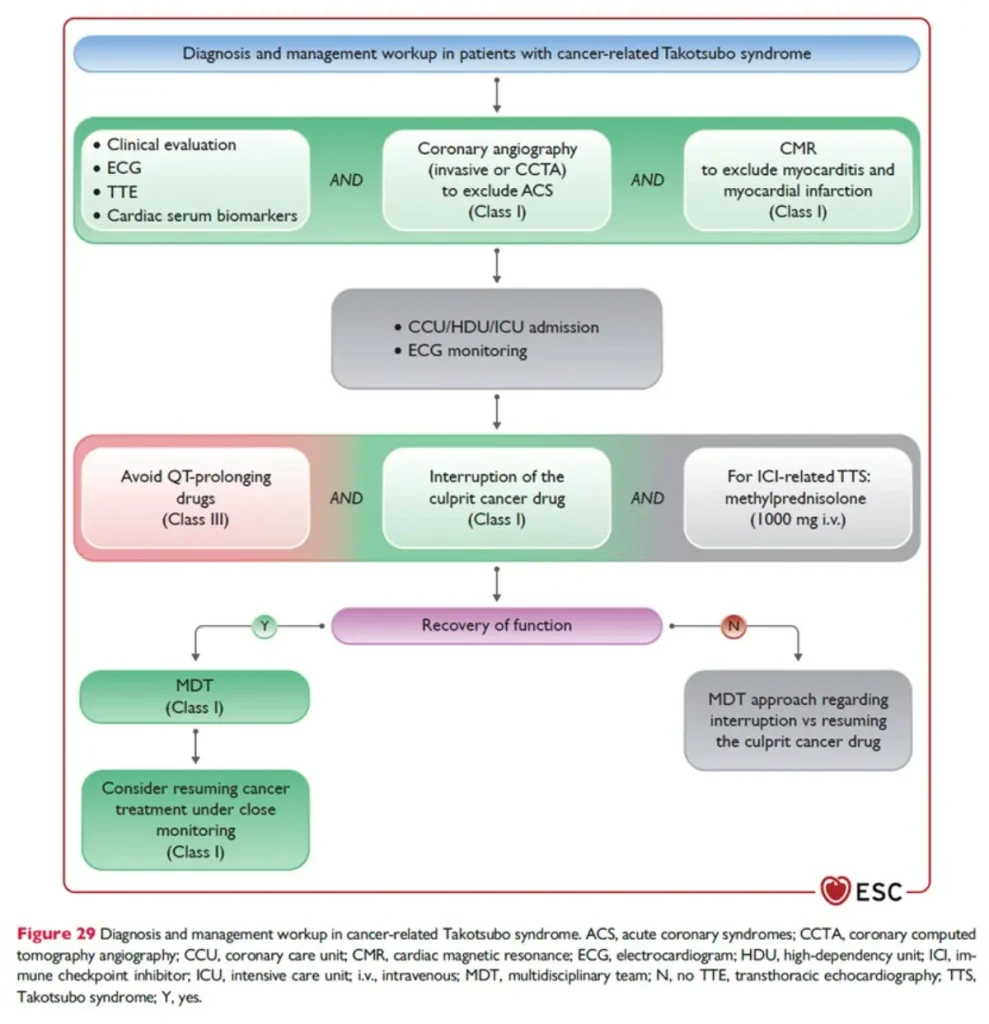
Therapy and oncology treatment
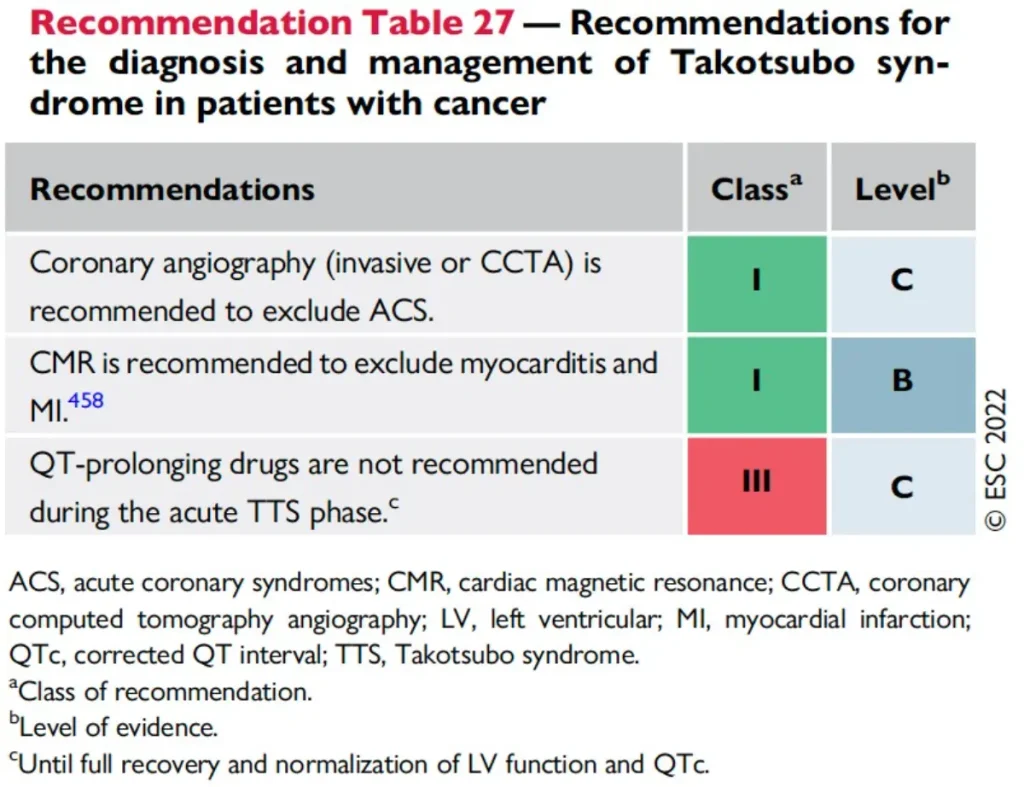
References
Lyon AR et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur Heart J (2022) : 00, 1-133.
| Chemotherapy | Carboplatin, cisplatin Asparaginase Cyclophosphamide Anthracyclines : epirubicin, doxorubicin, daunorubicin Antimetabolites : 5-fluorouracil, capecitabine, cytarabine, 6-mercaptopurine, fludarabine, methotrexate, gemcitabine, pemetrexed Irinotecan Taxanes : paclitaxel, docetaxel, cabazitaxel |
| Endocrine therapy | Tamoxifen |
| Targeted therapy | CDK-inhibitors : palbociclib, abemaciclib, ribociclib IMiDs : thalidomide, lenalidomide, pomalidomide Proteasome inhibitors : carfilzomib Angiogenesis-inhibitors : bevacizumab, axitinib, lenvatinib, pazopanib, sorafenib, sunitinib |
| Immune therapy | Immune checkpoint inhibitors CAR-T cell therapy |
| Other factors in cancer patients | Erythropoiesis-stimulating agents Central venous catheter <6mo after diagnosis |
+ consider cancer type and stage
Prevalence of CAT by cancer type
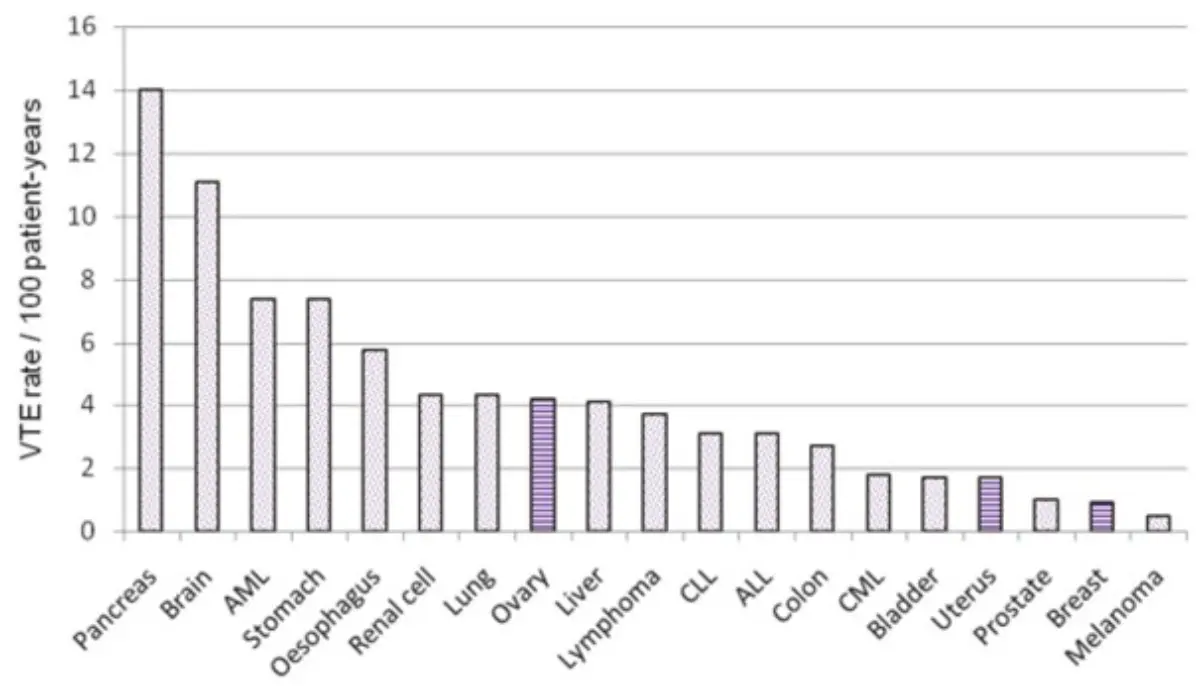
Diagnostic approach
The same diagnostic approach and monitoring of VTE (DVT & PE) as in non-cancer patients apply.
DIAGNOSTIC CAVEATS :
| Aspect | Key points |
| Incidental VTE / PE | Treat identical to symptomatic VTE / PE |
| PAC-associated thrombosis | Catheter may remain if functional, non-infected, well-positioned, and still indicated; otherwise consider removal (discuss with oncologist). After removal: therapeutic anticoagulation for 3 months. If left in place: extended therapeutic anticoagulation |
| Choice of anticoagulant | DOACs (edoxaban, rivaroxaban, apixaban) are non-inferior to LMWH; avoid vitamin K antagonists (VKA) |
| Treatment duration | Minimum 6 months Consider sufficiently long anticoagulant therapy + imaging reassessment |
| Extended treatment – therapeutic dose | Continue therapeutic anticoagulation in active malignancy, ongoing cancer therapy, or metastatic disease |
| Extended treatment – secondary prevention | Consider reduced-dose apixaban for long-term prophylaxis in selected patients |
| IVC filter | Use in CAT remains controversial |
| Aspect | Key points |
| Very high bleeding risk | Active/recent (<1 month) major bleeding; recent/evolving intracranial lesions; platelets <25,000/mm³ |
| Conditions favouring LMWH | Unoperated GI/GU cancer; GI comorbidities or toxicity; severe renal dysfunction (CrCl <15 mL/min); major drug–drug interactions with DOACs; platelets <50,000/mm³ |
| Thrombocytopaenia (<50,000/mm³) | Multidisciplinary discussion required; consider dose-reduced LMWH |
| Primary VTE prophylaxis | Evidence strongest in post-operative setting: prophylactic LMWH up to 4 weeks postoperatively |
| Cancer therapy interruption | Generally not required in VTE unless in severe PE/VTE. |
| Severe VTE / ATE in IMID therapy / angiogenesis inhibition | Discuss alternatives with treating oncologist |
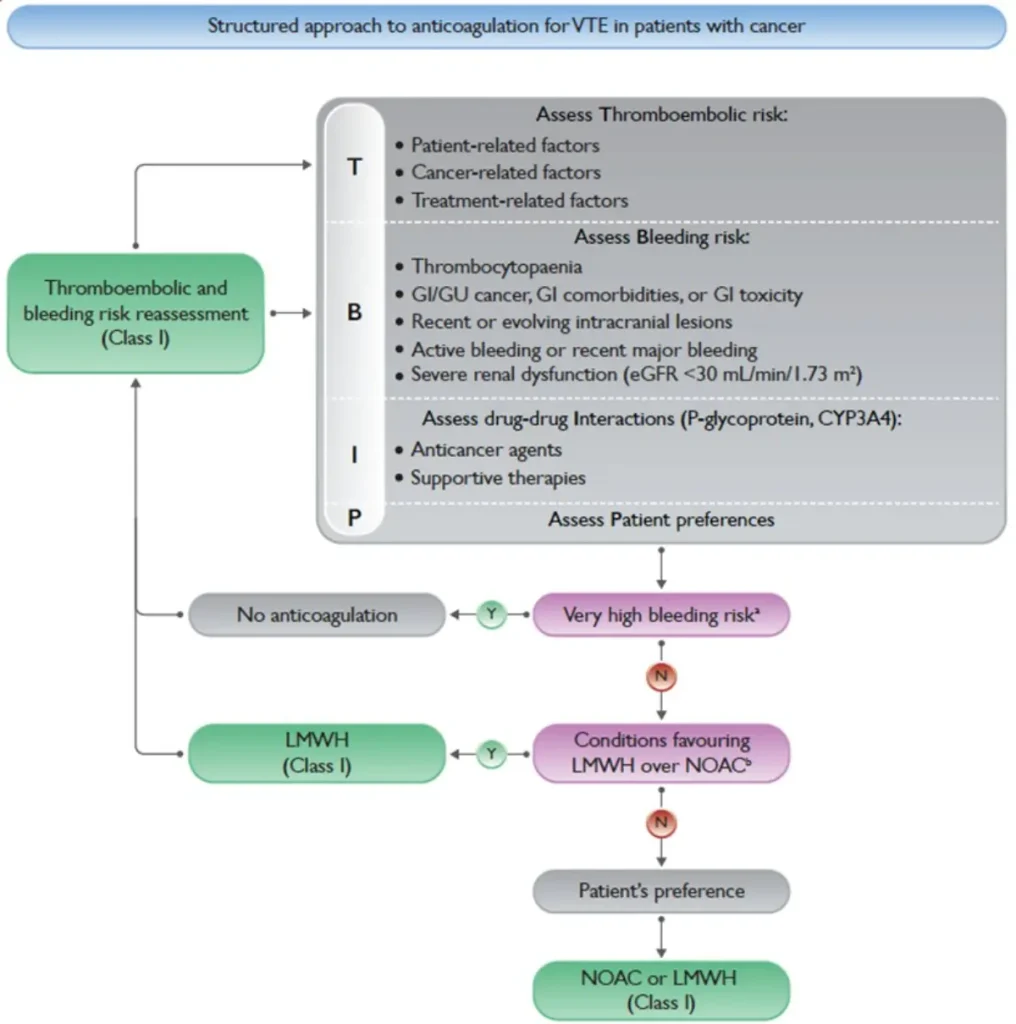
References
Lyon AR et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur Heart J (2022) : 00, 1-133.
Gevaert SA et al. Evaluation and management of cancer patients presenting with acute cardiovascular disease: a Clinical Consensus Statement of the Acute CardioVascular Care Association (ACVC) and the ESC council of Cardio-Oncology—part 2: acute heart failure, acute myocardial diseases, acute venous thromboembolic diseases, and acute arrhythmias. Eur Heart J Acute Cardiovasc Care (2022) : 11, 865-74.
Mahé I, Carrier M, Didier M, et al. Extended reduced-dose apixaban for cancer-associated venous thromboembolism. NEJM. Published online March 29, 2025.
Bauersachs RM et al. Guidelines for the management of cancer and thrombosis – Special aspects in women, Thrombosis Research, Volume 135, Supplement 1, 2015,
Cancer-therapy related cardiac dysfunction
Encompasses cardiac injury, cardiomyopathy and heart failure[i].
Can be symptomatic of asymptomatic
1. Symptomatic CTRCD
HF syndrome with symptoms and clinical signs of volume overload and/or inadequate perfusion caused by structural and/or functional cardiac abnormalities
2. Asymptomatic HF
More frequent in cancer patients then symptomatic HF
Grading is based on the LVEF change; LV GLS and/or biomarkers help further determine severity
| Symptomatic CTRCD | Very severe | HF requiring inotropic support, mechanical circulatory support or consideration of heart transplantation |
| Severe | HF hospitalization | |
| Moderate | Need for outpatient intensification of diuretic and HF therapy | |
| Mild | Mild HF symptoms, no intensification of therapy neede | |
| Asymptomatic CTRCD | Severe | New LVEF reduction to <40% |
| Moderate | New LVEF reduction by ≥10 percent points to LVEF 40-49% OR New LVEF reduction by <10 percent points to LVEF 40-49% AND new decline LV GLS >15% from baseline OR new rise in cardiac biomarkers | |
| Mild | LVEF ≥50% AND new decline LV GLS >15% from baseline AND/OR new rise in cardiac biomarkers |
Myocarditis
Myocarditis is an inflammatory disease of the heart; in the case of cancer patients mostly due to direct toxicity or to an immune-mediated event.
| Pathohistological diagnosis (EMB) | Multifocal inflammatory cell infiltrates with overt cardiomyocyte loss by light microscopy. |
| Clinical diagnosis#& | cTN elevation (new or significant change from baseline) with 1 major criterion or 2 minor criteria after excluding ACS and acute infectious myocarditis based on clinical suspicion. Major criterion:
Minor criteria:
Suggestive CMR findings. |
| Severity of myocarditis |
|
| Recovery |
|
# Clinical diagnosis should be confirmed with CMR of endomyocardial biopsy if possible and wiothout causing delays of treatment
& In a patient that is unwell, treatment with immunosuppression should be initiated promptly while awaiting further confirmatory testing
Vascular toxicities
Vacular toxicity is the induction or aggravation of vascular disease due to cancer therapy
Can be symptomatic of asymptomatic.
A. Asymptomatic vascular toxicities
recognized by changes in diagnostic testing parameters beyond what can be expected based on analytical and biological variability and common thresholds for abnormality.
B. Symptomatic vascular toxicities
Defined by societal guidelines.
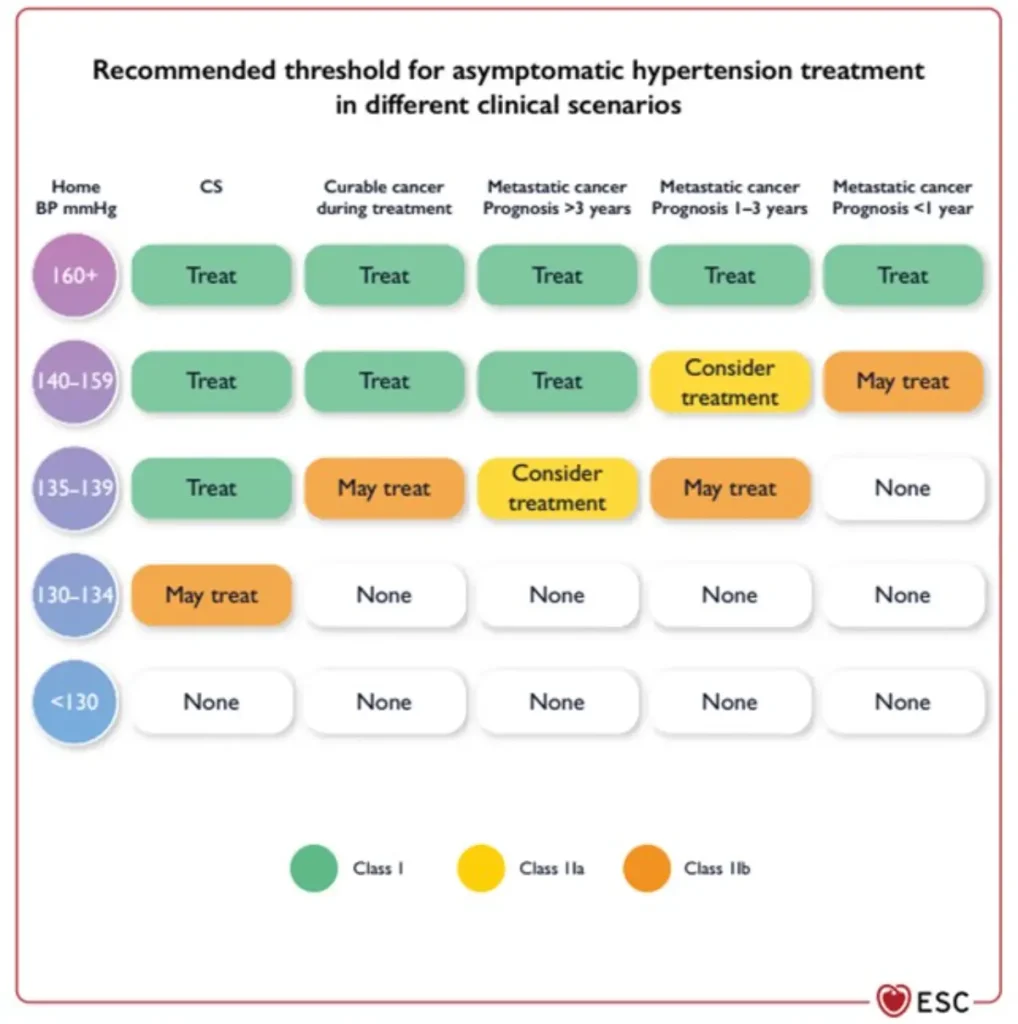
C. Hypertension
An increase in systolic and/or diastolic blood pressure (BP) after initiation of cancer therapy, without any other contributing changes.
Different agents may have variable hypertensive effects and there is remarkable inter-individual variation.
Confirm diagnosis with out-of-office BP measurements.
| Category | Criteria |
| Normal | SBP ≤130 mmHg and DBP ≤80 mmHg |
| Threshold to initiate AH treatment before, during and after cancer therapy | CVD or ASCVD ≥10%: SBP ≥130 mmHg and/or DBP ≥80 mmHg Otherwise: SBP ≥140 mmHg and/or DBP ≥90 mmHg |
| Threshold to withhold cancer therapy | SBP ≥180 mmHg and/or DBP ≥110 mmHg |
| Exaggerated hypertensive response | Systolic BP increase >20 mmHg or mean BP increase >15 mmHg |
| Hypertensive emergency | (Very high) BP elevations associated with acute hypertension-mediated organ damage (heart, retina, brain, kidneys, large arteries), requiring immediate BP reduction to limit extension or promote regression of target organ damage |
QTc prolongation
Fridericia formula (QTcF = QT/RR¹ᐟ³) is preferred for QT correction in cancer patients, as it reduces the overestimation of QT prolongation seen with Bazett’s formula — particularly at higher heart rates — and minimizes inappropriate management changes during QT-prolonging cancer therapy.
| QTcF <480ms | Acceptable: continue current treatment |
| QTcF 480-500ms | Prolonging: proceed with caution; minimize other QT prolonging medications; replete electrolytes |
| QTcF >500ms | Prolonged: stop treatment and evaluate. May require dose reduction or alternative treatment |
References
Lyon A et al (2022) 2022 ESCGuidelines on cardio-oncology developed in collaboration with the European HematologyAssociation(EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS)
Herrmann J et al. Defining cardiovascular toxicities of cancer: an International Society of Cardio-Oncology (IC-OS) consensus statement. Eur. Heart J. 2022; 43:280-299.
Richardson DR et al. Association of QTc Formula With the Clinical Management of Patients With Cancer. JAMA Oncol. 2022;8(11):1616–1623. doi:10.1001/jamaoncol.2022.4194
According to the Society for Cardiovascular Angiography & Interventions (SCAI) 2016 expert consensus document, the following rules of thumb apply:
References
Indications:
| Main cardiovascular complications | Key clinical considerations | |
| Pre-HSCT (baseline assessment) |
| |
| Early phase (<100 days post-HSCT) |
|
|
| Intermediate phase (3–12 months) |
|
|
| Late phase (>1 year post-HSCT) |
|
|
| GVHD-related complications (all phases) | Acute GVHD: myocarditis, heart failure, arrhythmias, conduction disorders, pericardial effusion, thrombosis. Chronic GVHD: |
|
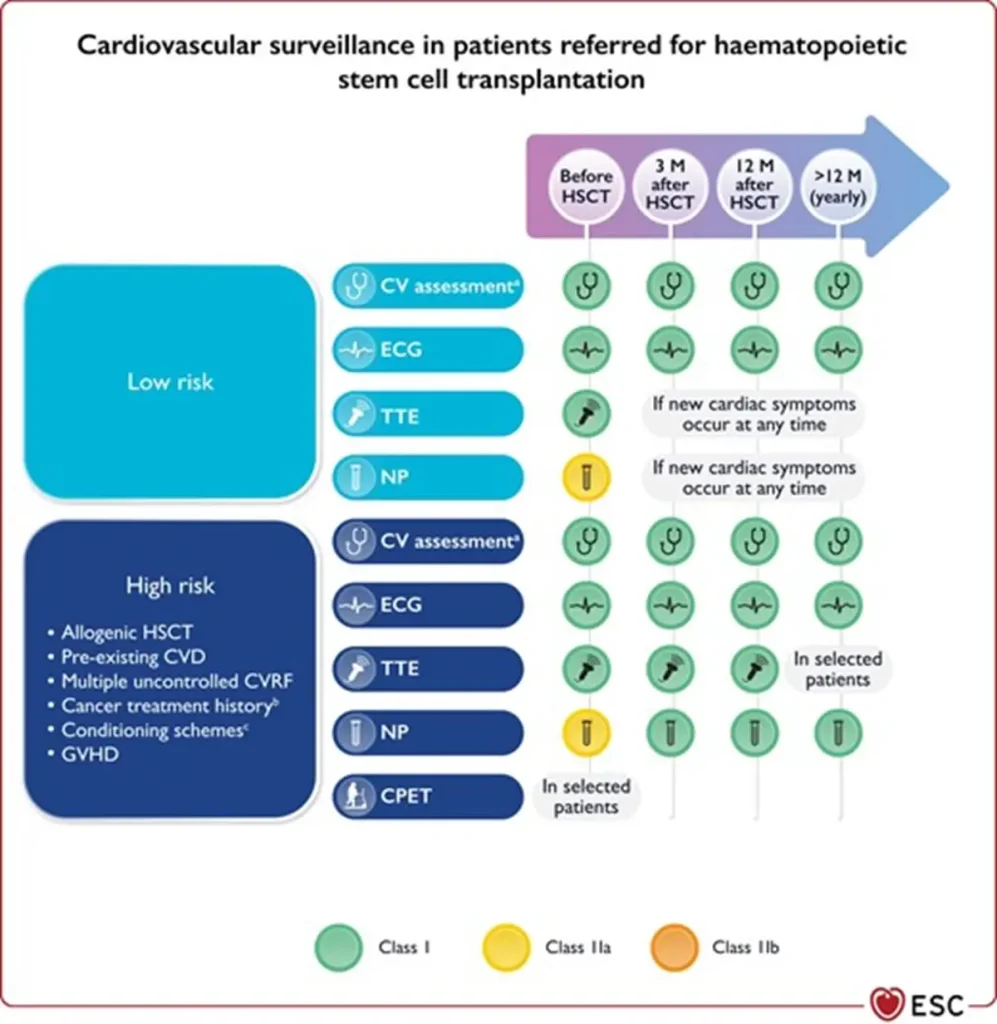
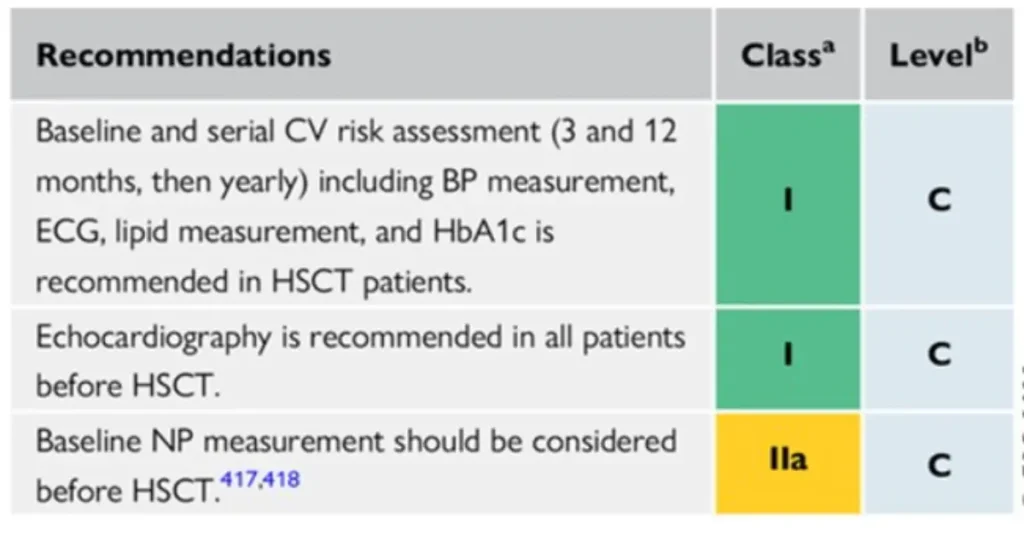
Footnote for figure: BNP, B-type natriuretic peptide; BP, blood pressure; CPET, cardiopulmonary exercise testing; CV, cardiovascular; CVD, CV disease; CVRF, cardiovascular risk factors; ECG, electrocardiogram; GVHD, graft vs. host disease; HbA1c, glycated haemoglobin; HSCT, haematopoietic stem cell transplantation; M, months; NP, natriuretic peptides (including BNP or NT-proBNP); NT-proBNP, N-terminal pro-BNP; TTE, transthoracic echocardiography. aIncluding physical examination, BP, lipid profile, and HbA1c. bMediastinal or mantle field radiation, alkylating agents, >250 mg/m2 doxorubicin or equivalent. cTotal body irradiation, alkylating agents.
References
| Agent | Main oncologic / hematologic indications | Type of cardiotoxicity | Estimated risk |
| Doxorubicin (Adriamycin®, Caelyx® (pegylated liposomal)) Daunorubicin Epirubicin Idarubicin | Solid tumors: Hematologic malignancies: |
| Symptomatic CTRCD:
Asymptomatic CTRCD:
|
| Mitoxantrone | Hematologic malignancies: Solid tumors: Non-oncologic indications: |
|
|
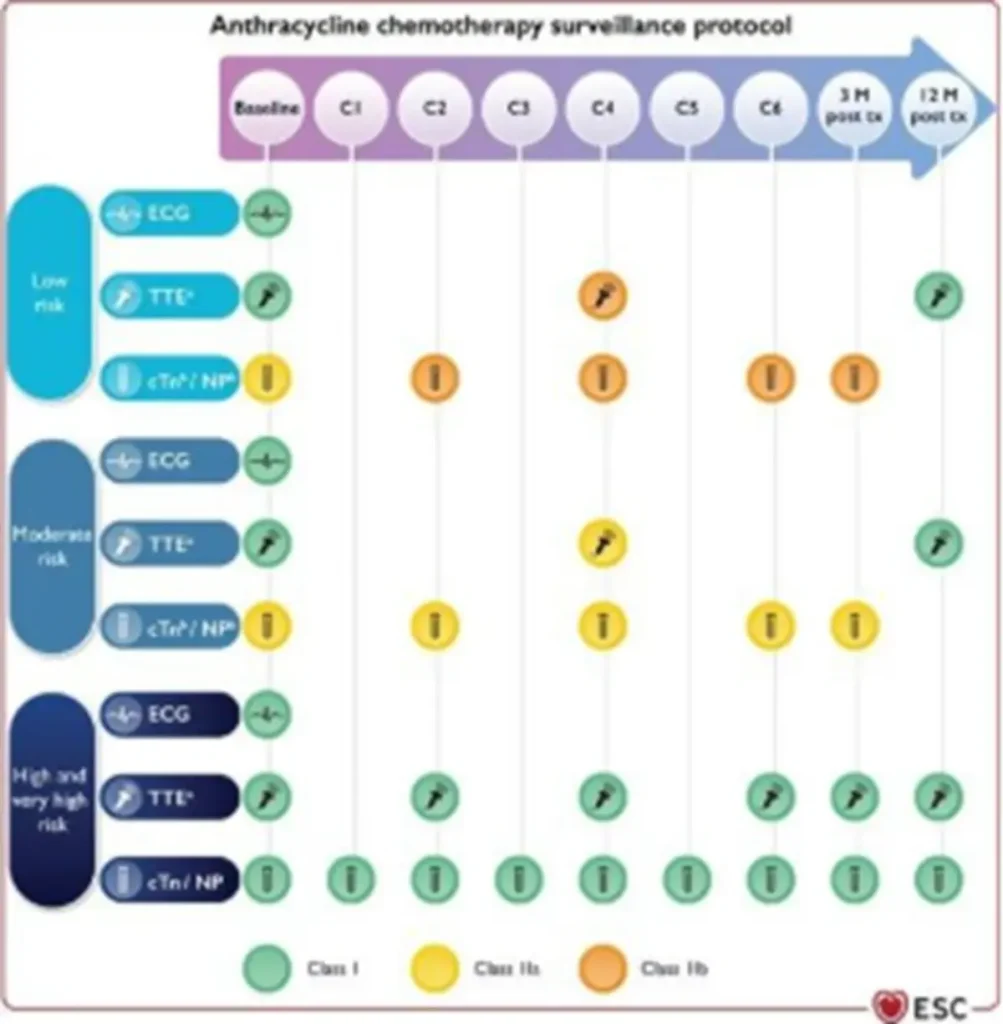
Proposed Surveillance (ESC guidelines):
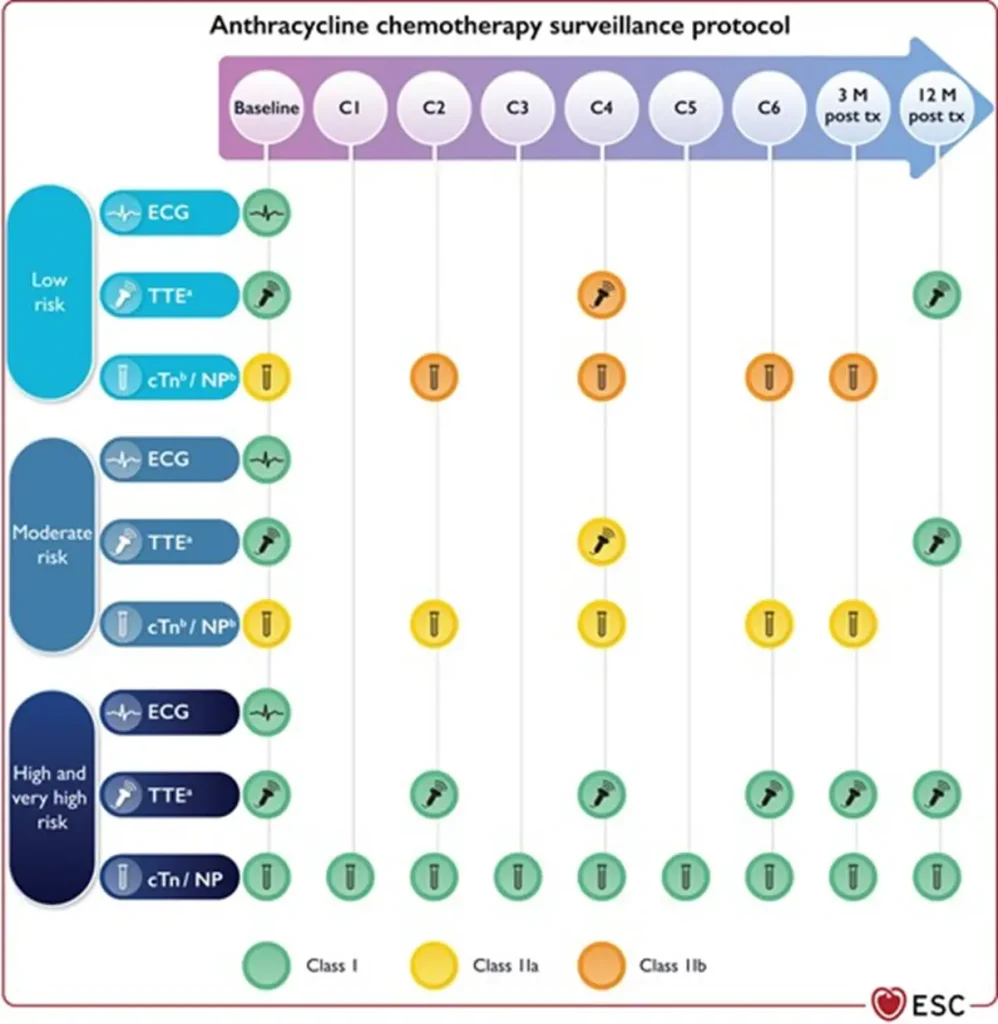
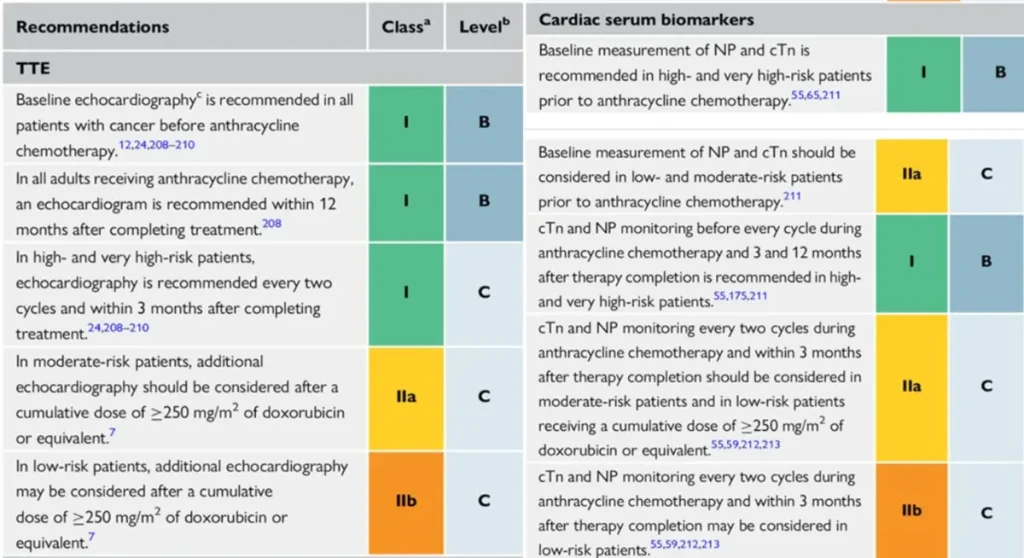
Of note: NTproBNP is not reimbursed in Belgium
Anthracycline equivalent dosis calculator: https://www.cancercalc.com/anthracycline.php
References
| Agent (commercial name) | Main oncologic indications | Type of cardiotoxicity | Estimated risk |
| Trastuzumab (Herceptin®) |
HER2-positive (IHC 3+ or FISH+) breast cancer (early and metastatic). HER2-positive gastric, gastro-esophageal junction, and selected pulmonary cancers. |
|
Symptomatic CTRCD:
|
| Pertuzumab (Perjeta®) |
HER2-positive breast cancer (early and metastatic), in combination with trastuzumab and chemotherapy. |
||
| Trastuzumab emtansine (T-DM1) (Kadcyla®) | HER2-positive breast cancer in the (neo)adjuvant and metastatic setting after prior trastuzumab/taxane therapy. |
LVEF decline: ~0.9% Symptomatic HF is uncommon |
|
| Trastuzumab deruxtecan (T-DXd) (Enhertu®) |
HER2-positive metastatic breast cancer. HER2-low (IHC 2+, FISH−) and HER2 ultra-low (IHC 1+) metastatic breast cancer. |
LVEF decline: ~4.2% Symptomatic HF is uncommon |
Across all HER2-targeted therapies, the risk of cancer therapy–related cardiac dysfunction (CTRCD) is significantly increased when combined with or preceded by anthracycline therapy, emphasizing the importance of baseline cardiovascular risk stratification and cardiac monitoring.
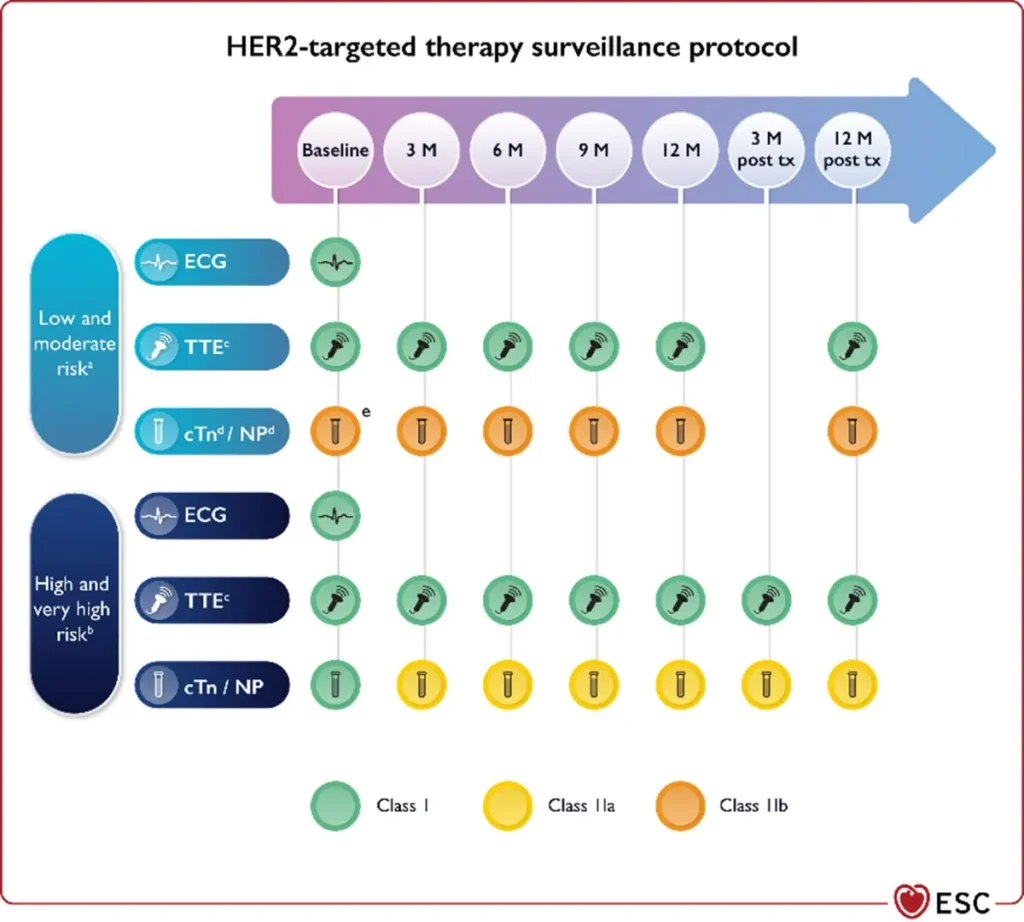
Proposed Surveillance (ESC guidelines):
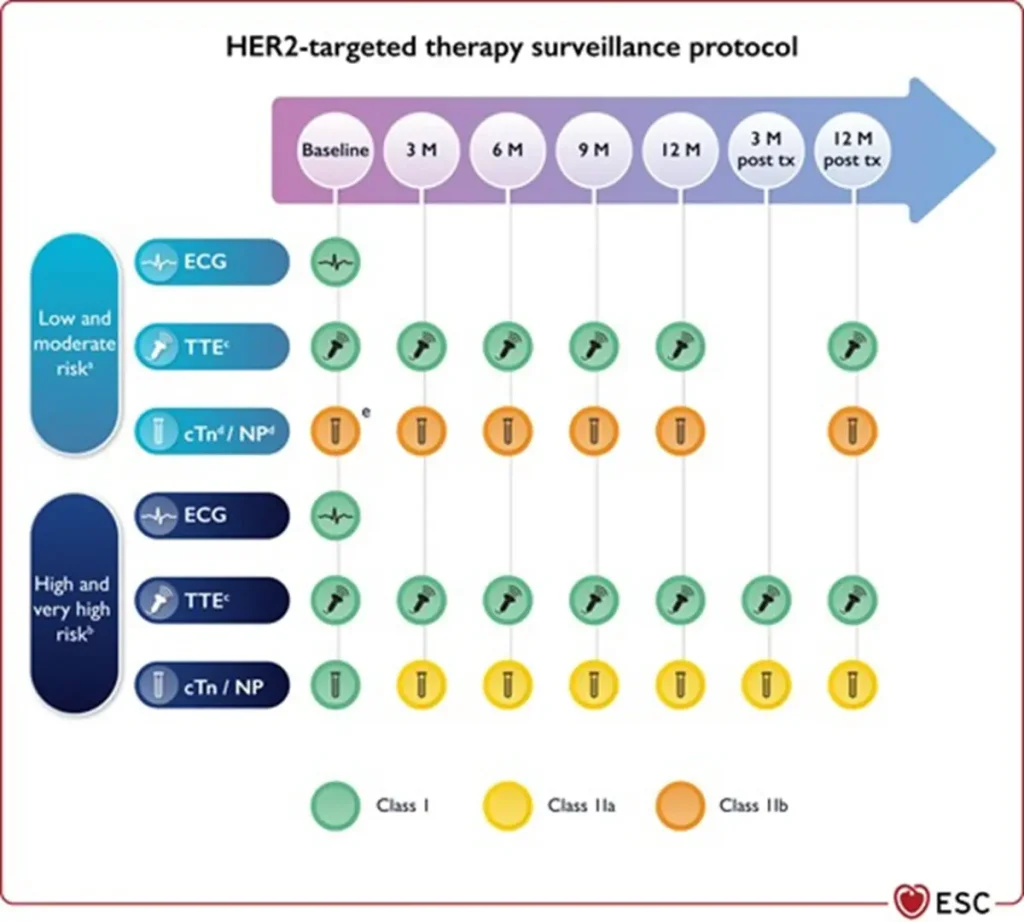
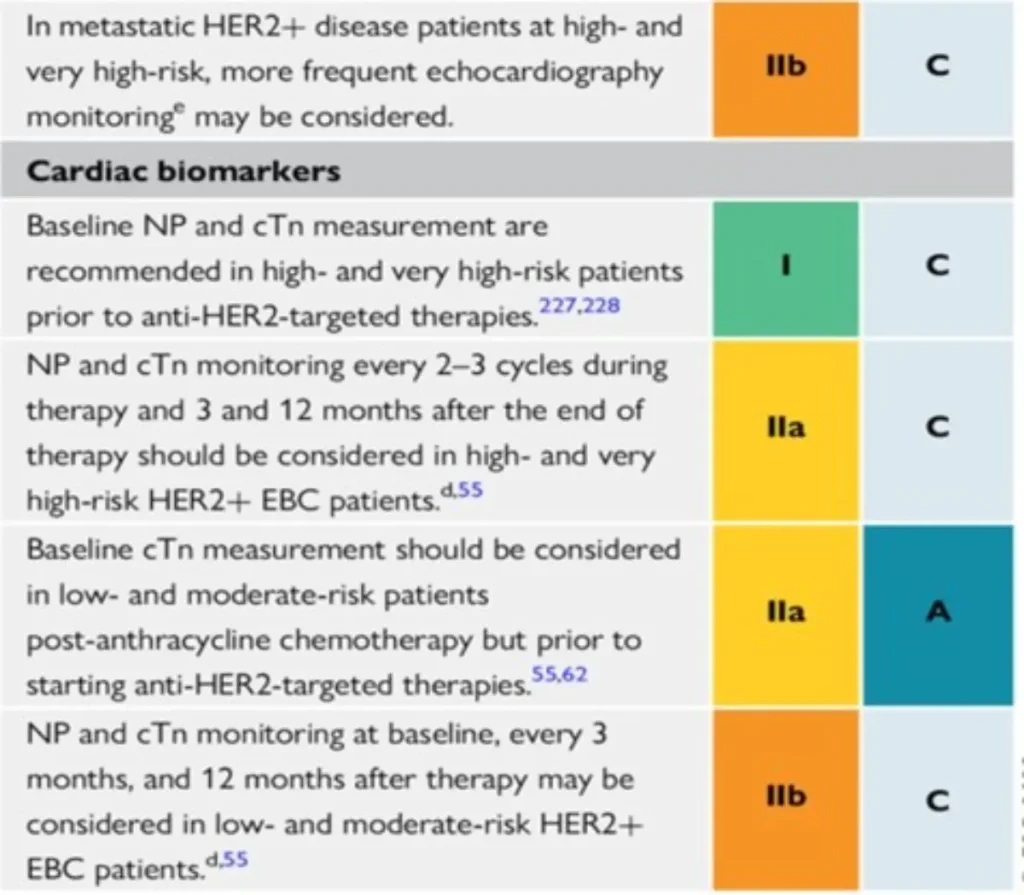
NTproBNP is not reimbursed in Belgium
References
| Agent | Main oncologic / hematologic indications | Type of cardiotoxicity | Estimated risk and risk factors |
| 5-Fluorouracil (5-FU) Capecitabine | Gastrointestinal cancers: colorectal, gastric, pancreatic, esophageal cancer.Also used in (neo)adjuvant regimens for head and neck cancers. |
|
Any cardiac event: ~3–7% in most series (reported range 0.99–19.9%). Most frequent are ischemic events. Risk factors: pre-existing CAD, prior chest radiotherapy, renal insufficiency, dihydropyrimidine dehydrogenase (DPD) deficiency. |
| Cytarabine | Hematological indications:
|
|
Pericarditis: <5%. Cardiomyopathy: rare, usually associated with high-dose regimens. |
| Methotrexate | Hematologic malignancies: Acute lymphocytic leukemia (ALL) (systemic and intrathecal), lymphomas (including primary CNS lymphoma). Solid tumors: osteosarcoma, head and neck cancers. Non-oncological indications: autoimmune disease and trophoblastic disease (low dose) | Pericarditis / effusion: <5%, often dose-related. Cardiomyopathy: uncommon. |
Proposed Surveillance for 5-Fluorouracil (5-FU) and Capecitabine (ESC guidelines):
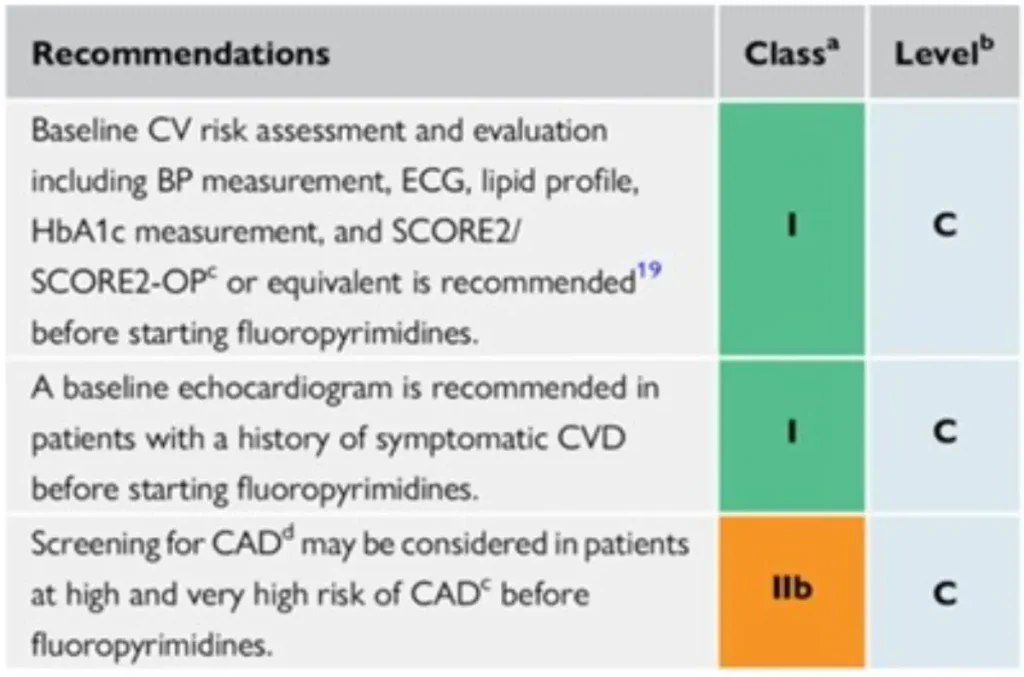
No specific surveillance recommendations for cytarabine, methotrexate : consider surveillance strategies for other anticancer drugs that are given in combination.
References
| Drug class / Agent | Main oncologic indications | ESC-aligned cardiotoxicity profile | Estimated risk |
| Anti-PD-1 / Anti-PD-L1 Nivolumab (Opdivo®) Pembrolizumab (Keytruda®) Atezolizumab (Tecentriq®) Durvalumab (Imfinzi®) Cemiplimab (Libtayo®) Avelumab (Bavencio®) Dostarlimab (Jemperli®) | Broad spectrum of solid tumours, including: Non-small cell lung cancer (NSCLC), melanoma, renal cell carcinoma, urothelial carcinoma, head and neck cancers, MSI-H / dMMR colorectal and other GI cancers; hepatocellular carcinoma, cervical and endometrial cancer, cutaneous squamous cell carcinoma (Indication depends on tumour type and PD-1/PD-L1 status.) | Immune-mediated CTR-CVT:
|
|
| Anti-CTLA-4 Ipilimumab (Yervoy®), Tremelimumab (Imjudo®) | Melanoma, hepatocellular carcinoma, renal cell carcinoma, selected GI and other solid tumors, often in combination with anti-PD-1 therapy. | ||
| Anti-LAG-3 Relatlimab (Opdualag® — fixed-dose combination with nivolumab) | Melanoma, typically in combination with nivolumab. |
Surveillance:
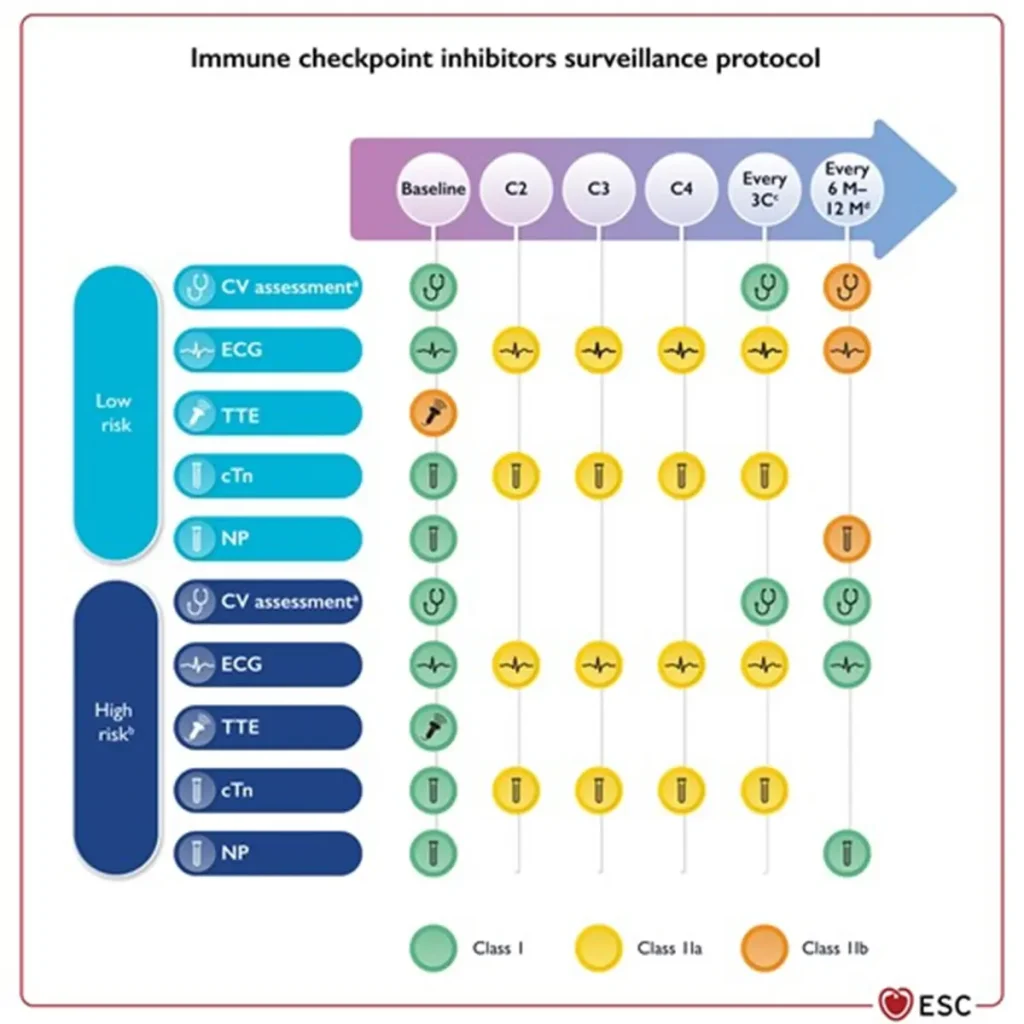
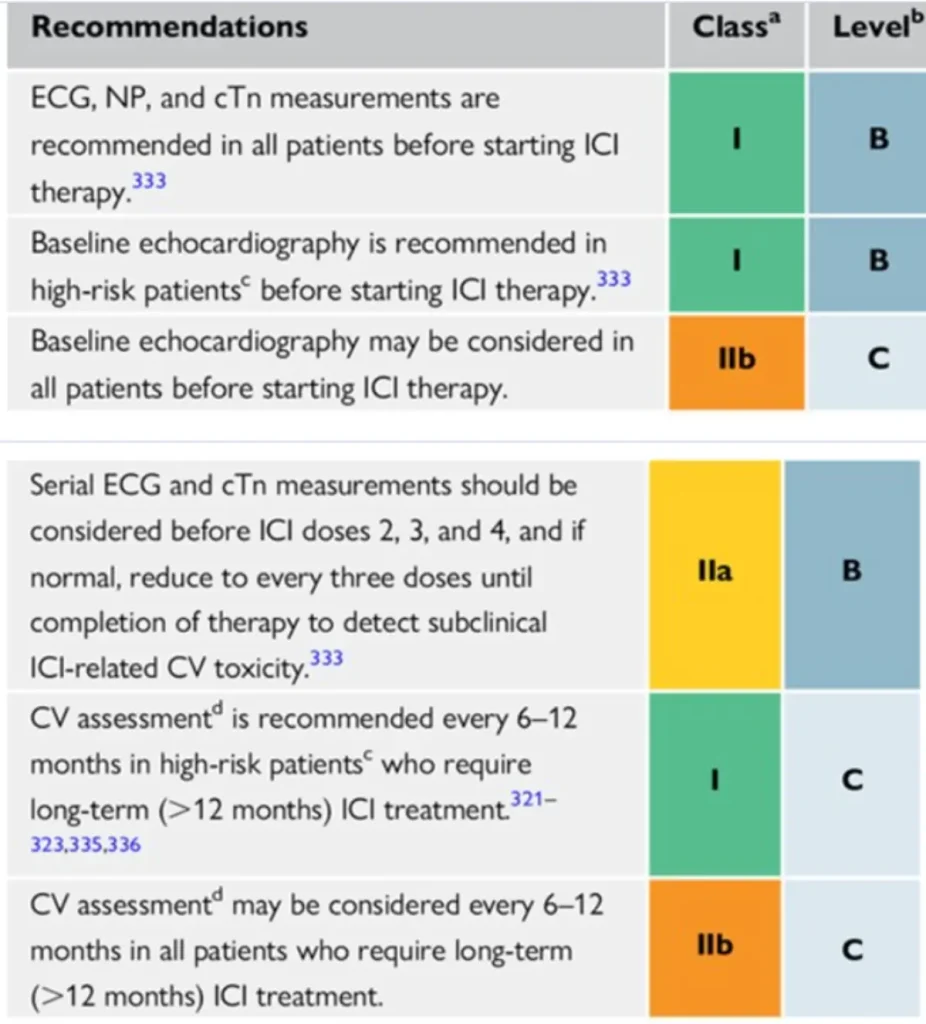
Consider multidisciplinary discussion via BSMO BITOX Immunomanager submission
https://bsmo.be/multidiscplin-immunotox-meeting/
References
| Therapy | Main oncologic indications | Type of cardiotoxicity | Approximate risk |
CAR T-cell therapy (CAR-T) (e.g. tisagenlecleucel, axicabtagene ciloleucel, lisocabtagene maraleucel, idecabtagene vicleucel, ciltacabtagene autoleucel): | Hematologic malignancies, depending on product:
| Hematologic malignancies, depending on product:
| LVEF or GLS decline: ~5–10% Symptomatic HF: 2–15% Acute coronary syndrome: 1.4–7% Any arrhythmia: 0.8–12.2% Atrial fibrillation: 0.4–7.6% Hypotension: up to 87% (often CRS-related) |
Tumor-infiltrating lymphocytes (TIL) | Advanced or metastatic cutaneous melanoma. Under investigation for other solid tumors. |
| Hypotension: ~2.6% |
Consider multidisciplinary discussion via BSMO BITOX Immunomanager submission
https://bsmo.be/multidiscplin-immunotox-meeting/
Proposed surveillance (ESC guidelines):
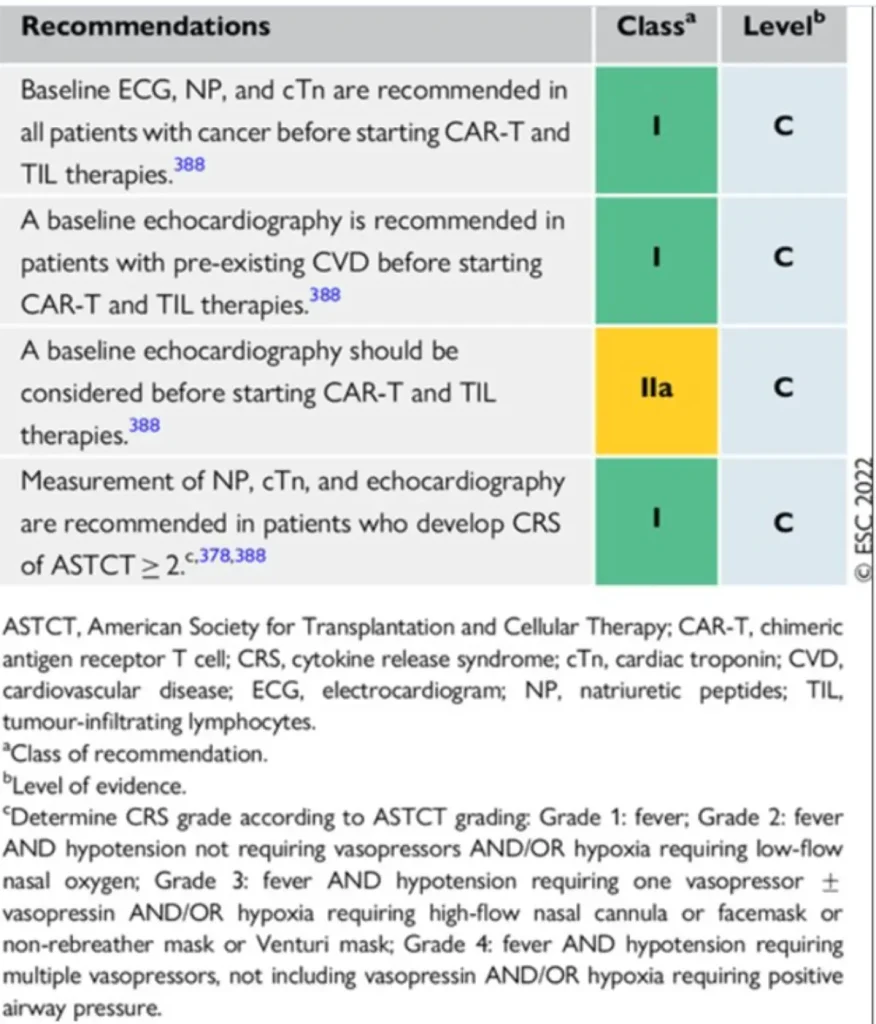
Grading of cytokine release syndrome Cytokine Release Syndrome (CRS) – ASTCT Consensus Criteria
| CRS Grade | Fever (≥38°C) | Hypotension | Hypoxia |
| Grade 1 | Present | None | None |
| Grade 2 | Present | Responsive to IV fluids (no vasopressors) | Requiring low-flow oxygen (≤6 L/min nasal cannula) |
| Grade 3 | Present | Requiring one vasopressor (± vasopressin) | Requiring high-flow oxygen (>6 L/min), face mask, or non-rebreather |
| Grade 4 | Present | Requiring multiple vasopressors (excluding vasopressin) | Requiring positive pressure ventilation (CPAP, BiPAP, or mechanical ventilation) |
References
Commonly / frequently implicated therapies
There is no safe heart dose!
| Pericardial disease | Acute RT-induced pericarditis (rare) Chronic pericarditis Constrictive pericarditis | Early Late Late |
| Coronary artery disease | Proximal fibrotic noncalcified stenoses | Late |
| Valvular heart disease | Aortic valve stenosis | Late Late Late |
| Conduction disease | Bradycardia +/- arrhythmias | Late |
| Myocardial disease | Diastolic dysfunction (myocardial fibrosis) | Intermediate – late |
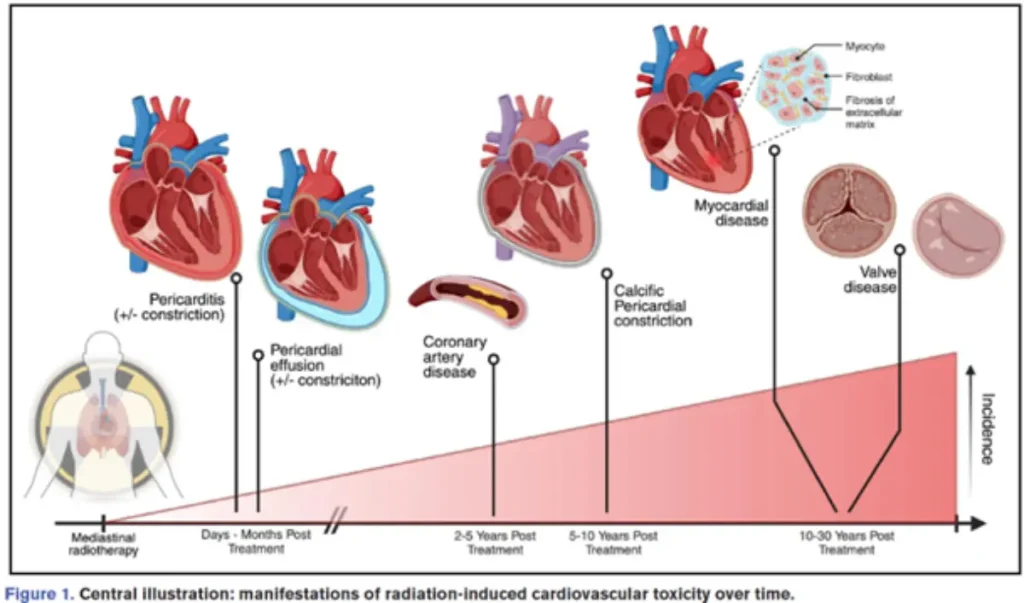
Diagnostic approach
Therapeutic approach : oncological aspects
Therapeutic approach : cardiovascular aspects
ESC Recommendations for baseline risk assessment of patients before radiotherapy to a volume including the heart
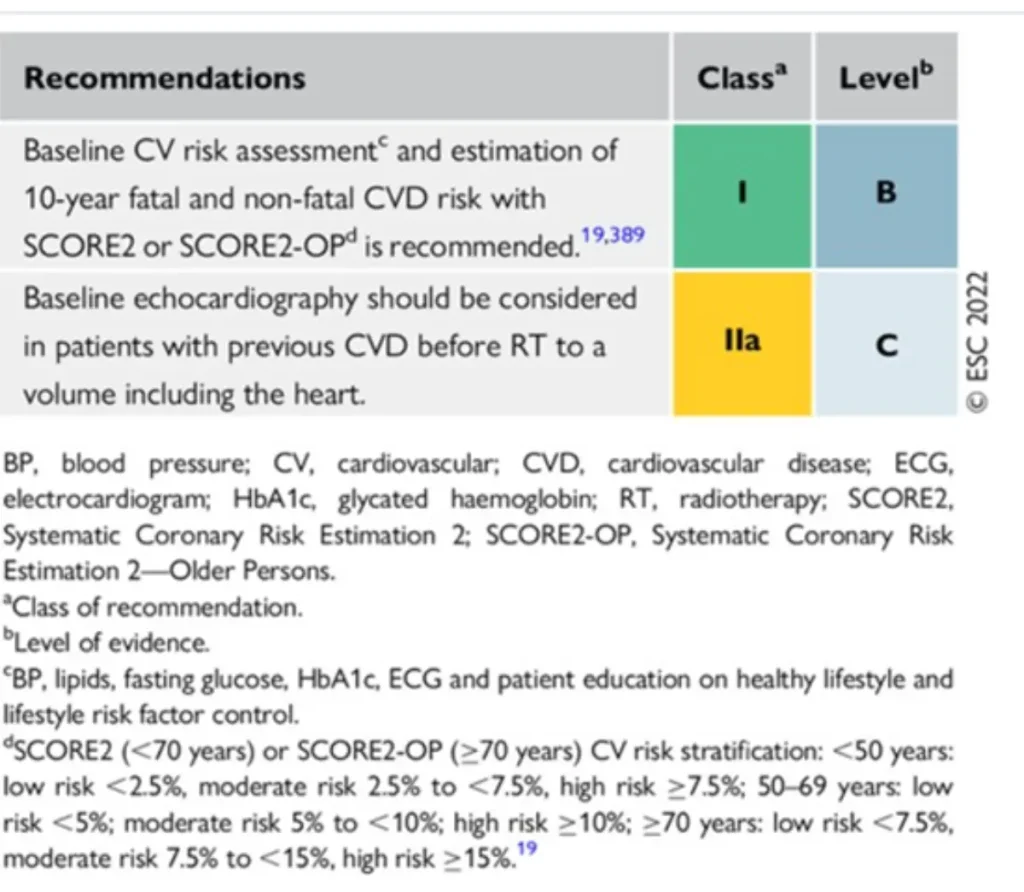
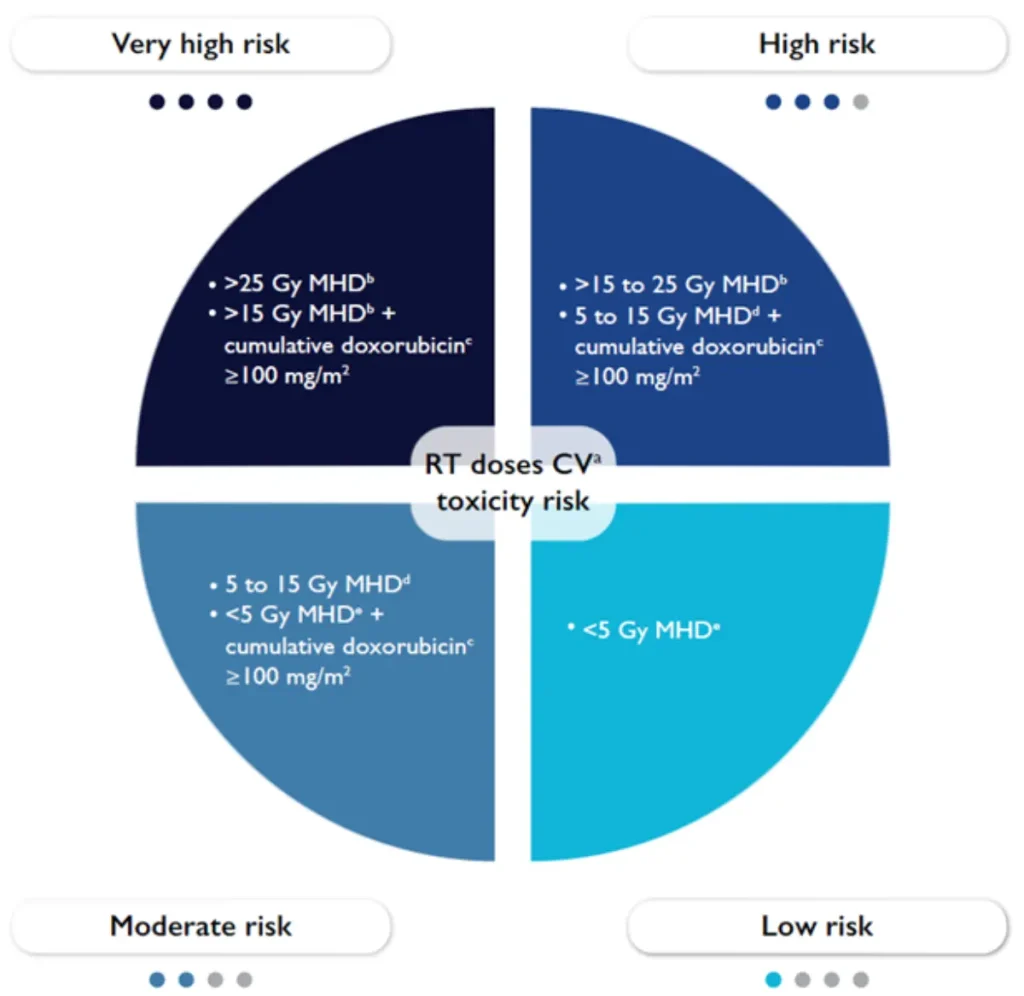
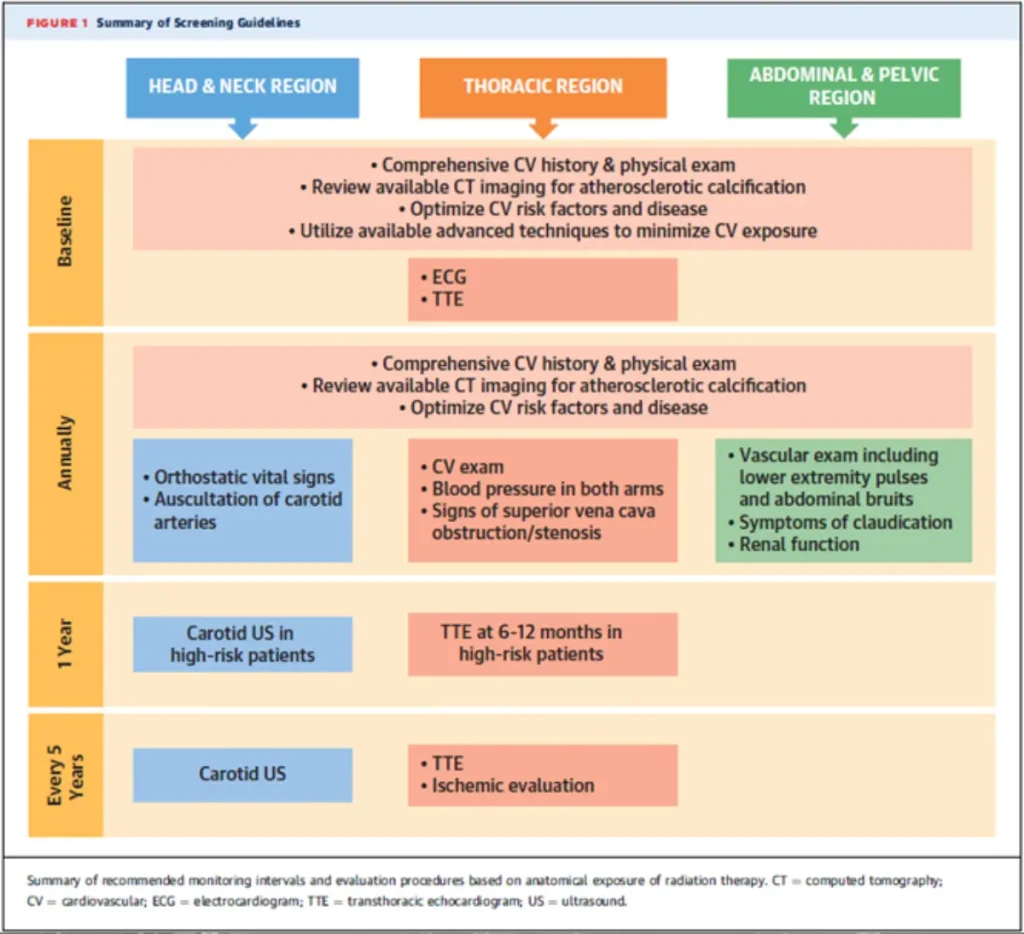
RT in patients with CIED
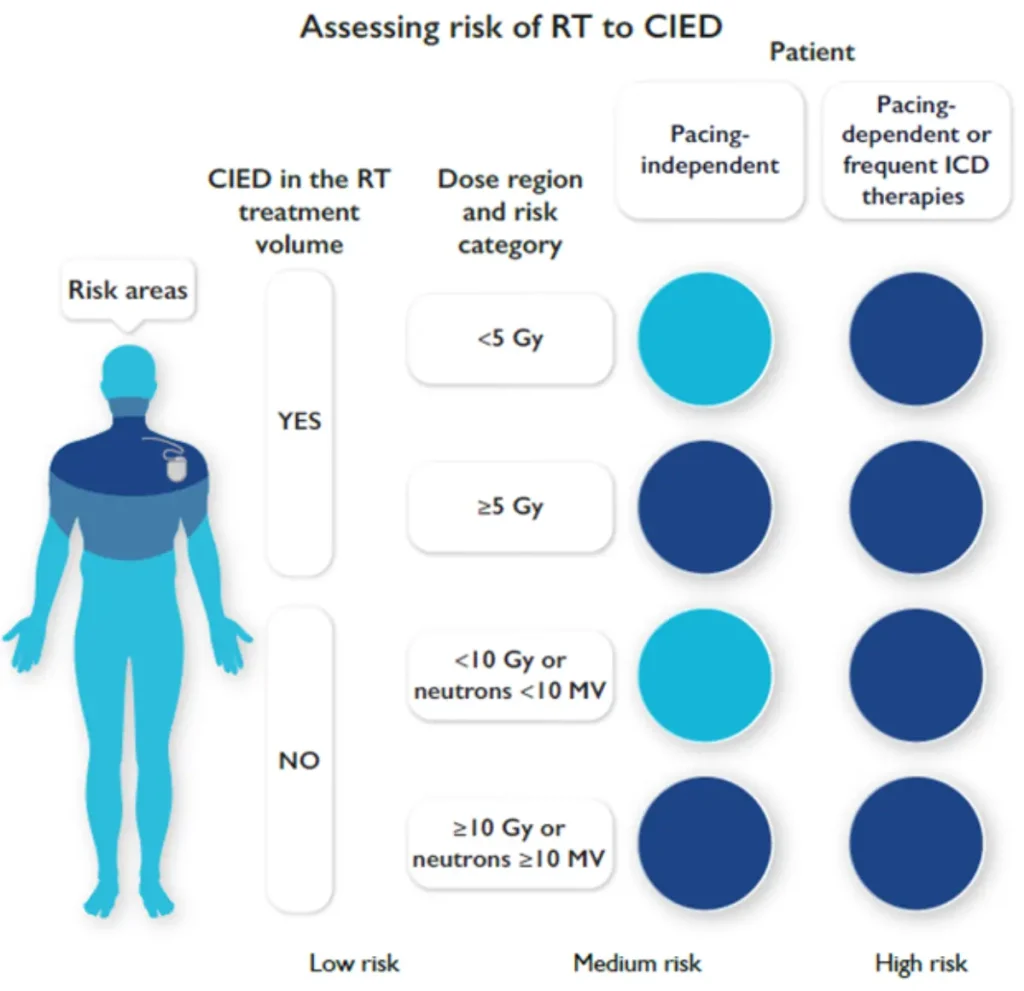
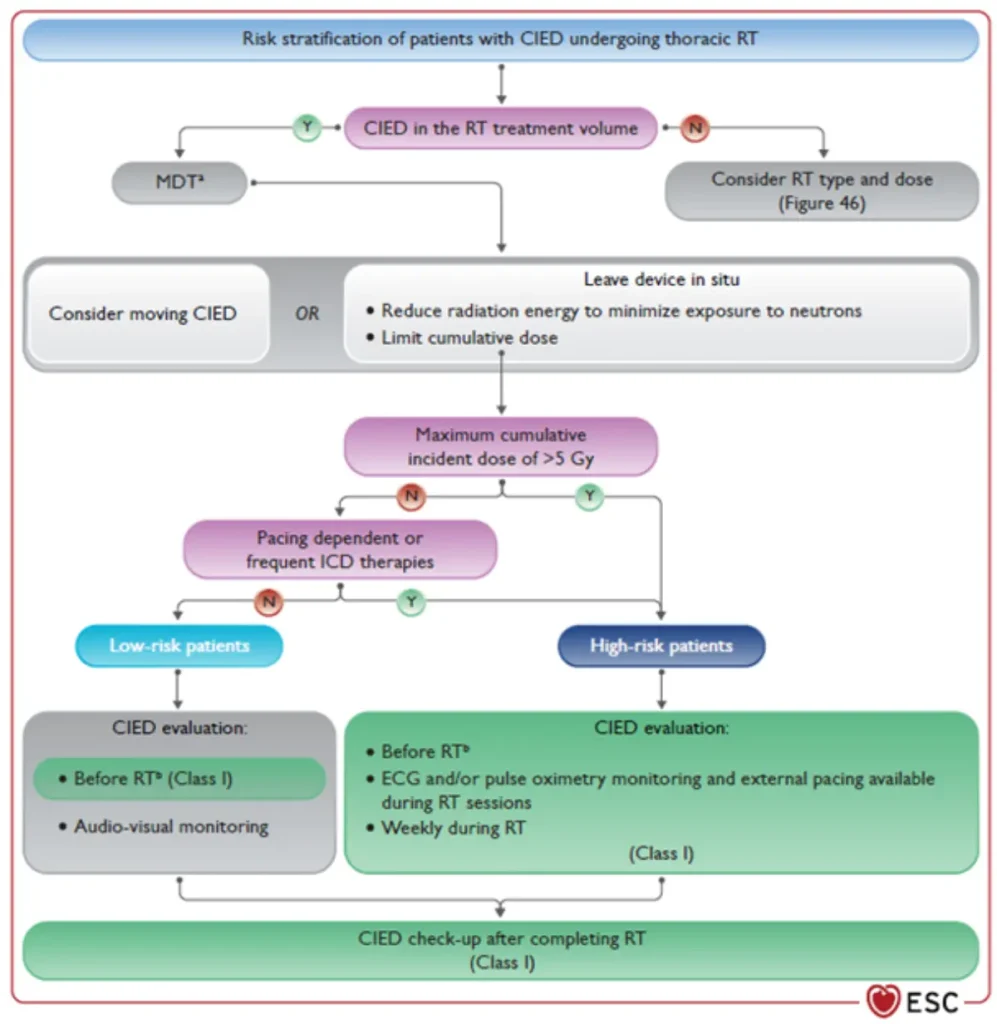
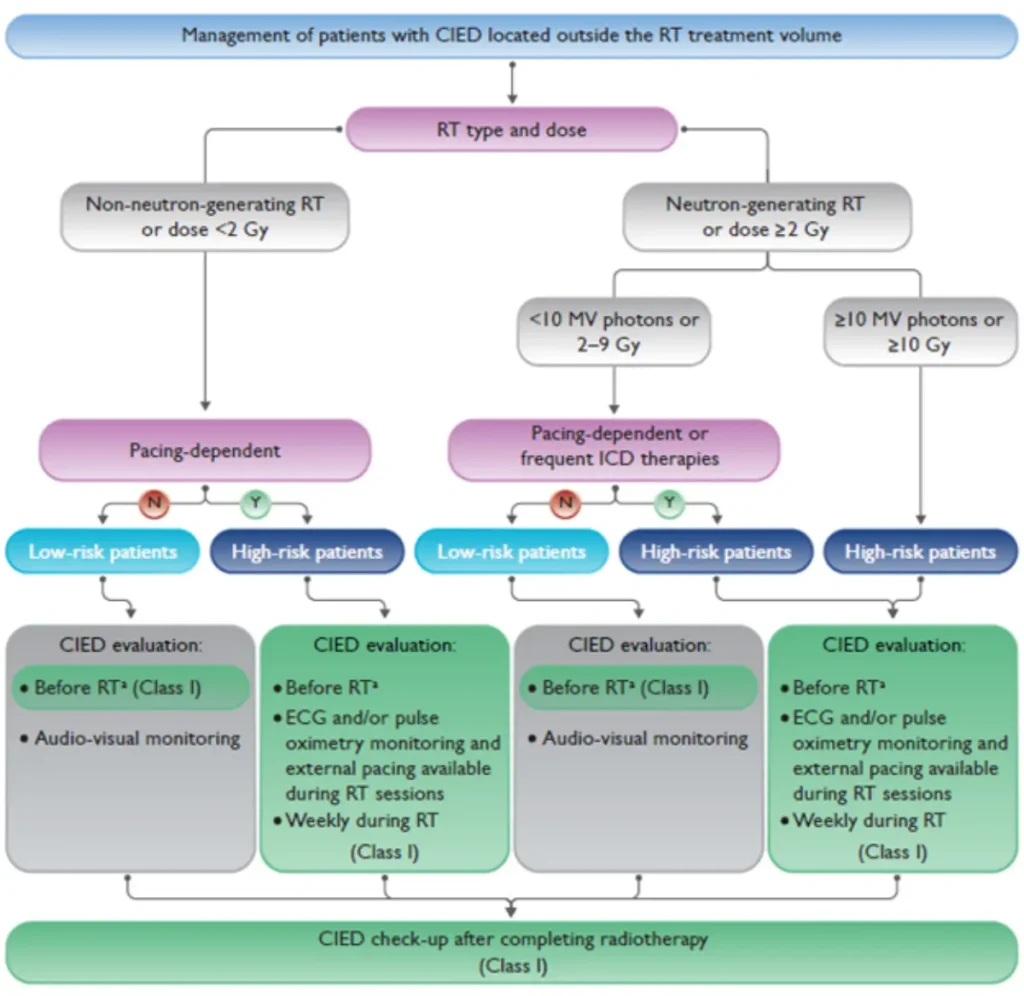
Radiotherapy (RT)–induced malfunction of cardiac implantable electronic devices (CIEDs) is rare. Higher risk with:
CIED behaviour within or near the RT treatment field remains unpredictable with potential RT-related CIED malfunctions:
Therefore, established precautionary measures should be followed to minimize patient risk (see figures below)
Risk stratification before planned radiotherapy in patients with CIED
References
Lyon AR et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur Heart J (2022) : 00, 1-133.
Wilson J, Hua CJ, Aziminia N, Manisty C. Imaging of the Acute and Chronic Cardiovascular Complications of Radiation Therapy. Circulation Cardiovasc Img (2025) : 18e017454.
Mitchell JD et al. State-of-the-art review. Cardiovascular Manifestations From Therapeutic Radiation. A Multidisciplinary Expert Consensus Statement From the International Cardio-Oncology Society. J Am Coll Cardiol CardioOnc (2021) : 3, 360-80.
| Agent | Main hematologic indications | Type of cardiotoxicity | Approximate risk |
| Thalidomide Lenalidomide Pomalidomide | Multiple myeloma (across different lines of therapy). Myelodysplastic syndromes with isolated deletion (5q) (lenalidomide). Selected other hematologic conditions. |
|
|
Surveillance:
no specific recommendation. Consider surveillance protocols for other anticancer drugs given in combination.
Alkylating agents
| Agents | Main oncologic / hematologic indications | Type of Cardiotoxicity | Approximate risk & notes |
| Cyclophosphamide, Ifosfamide | Hematologic indications:
Solid tumors:
• bone and soft-tissue sarcomas. | Acute/subacute CTR-CVT:
• Cardiomyopathy / heart failure | High-dose regimens (>120–150 mg/kg): cardiotoxicity ~8–20% (adults), ~5% (children). HF: reported <5% up to 10–29%. Onset typically 48 h to 10 days after exposure. |
| Cisplatin | Testicular, head & neck, lung, bladder, ovarian, and GI cancers (esophageal, gastric). |
| Considered low in monotherapy, higher (1-6%) when combined with other cardiotoxic agents (e.g. 5-FU). |
| Busulfan, Carmustine, Mitomycin, Melphalan | Hematologic indications:
Solid tumors: | Often in high-dose settings for stem cell transplantation:
• Pericardial effusion | Overall cardiac toxicity ~1–5% in transplant settings; data mainly from case series. |
Surveillance:
Antimicrotubule Agents – Taxanes and Vinca Alkaloids
| Agents | Main indications | Type of Cardiotoxicity | Approximate risk |
| Taxanes Paclitaxel, Docetaxel, Cabazitaxel | Breast, ovarian, lung, prostate, head & neck, gastric and esophageal cancers. |
|
Bradycardia: ~5–30% (often asymptomatic). Arrhythmias: <5%. |
| Vinca alkaloids Vincristine, Vinblastine | ALL, Hodgkin and non-Hodgkin lymphomas, multiple myeloma, pediatric solid tumors. | Rarely myocardial ischemia/infarction, arrhythmias, or hypertension. | Low cardiotoxicity risk: Cardiac events <1–2%. |
Surveillance:
Other Anticancer Agents
| Agents / Class | Main indications | Type of Cardiotoxicity | Approximate risk & notes |
| Arsenic trioxide | Acute promyelocytic leukemia (APL). | QT prolongation → torsades de pointes | QTc >470 ms: 20–30%. QTc >500 ms: 10–15%. TdP very rare (<1%) with monitoring. |
| Bleomycin | Hodgkin lymphoma, testicular and germ-cell tumors. | Rare myocardial ischemia, Raynaud phenomenon | <1–2% in preexisting cardiovascular disease or with combination therapy. |
| Interferon-α | Myeloproliferative neoplasms, hairy cell leukemia; historical use in CML, melanoma, RCC. | Arrhythmias, cardiomyopathy, heart failure, ischemia, hypertension | Cardiac events 5–15%; Heart failure <5%. |
| Tretinoin (ATRA) | APL (with arsenic or chemotherapy). | Not common, but can contribute to “differentiation syndrome” which may involve heart failure and pericardial effusion. | Differentiation syndrome with cardiac manifestations <5%. |
| Amsacrine | Relapsed/refractory AML. | Arrhythmias, QT prolongation; rare HF | Arrhythmias 5–10%; Heart failure <1%. |
| HDAC inhibitors Romidepsin, Vorinostat | Cutaneous and peripheral T-cell lymphomas. | QT prolongation, arrhythmias | QT prolongation: 10–20% (romipdesin) QTc >500 ms: ~1–5%. |
| Somatostatin analogs Lanreotide, Octreotide | Neuroendocrine tumors; hormonal symptom control. | Bradycardia (usually asymptomatic) | Bradycardia 5–15%, <1–2% clinically significant. |
Surveillance:
References
| Agent | Main hematologic indications | Type of cardiotoxicity | Approximate risk |
Bortezomib (Velcade®) | Multiple myeloma (induction, consolidation, maintenance). Mantle cell lymphoma; Occasional use in other plasma-cell or lymphoproliferative disorders. |
| Less frequent than carfilzomib
|
Carfilzomib (Kyprolis®) | Relapsed/refractory multiple myeloma, typically in combination regimens (e.g. lenalidomide/dexamethasone and other) |
|
|
Multiple myeloma drug-related cardiovascular toxicities

Footnote: AF, atrial fibrillation; ATE, arterial thromboembolism; DM, diabetes mellitus; EMA, European Medicines Agency; FDA, Food and Drug Administration; HF, heart failure; HG, hyperglycaemia; HTN, hypertension; MedDRA, medical dictionary for regulatory activities; MI, myocardial infarction; PH, pulmonary hypertension; VTE, venous thromboembolism. Adverse reactions reported in multiple clinical trials or during post-marketing use are listed by system organ class (in MedDRA) and frequency. If the frequency is unknown or cannot be estimated from the available data, a blank space has been left. aIxazomib produces peripheral oedema in up to 18% of patients and hyperglycaemia in combination with lenalidomide or pomalidomide and dexamethasone.
Proposed Surveillance during therapie with proteosome inhibitors (ESC guidelines):
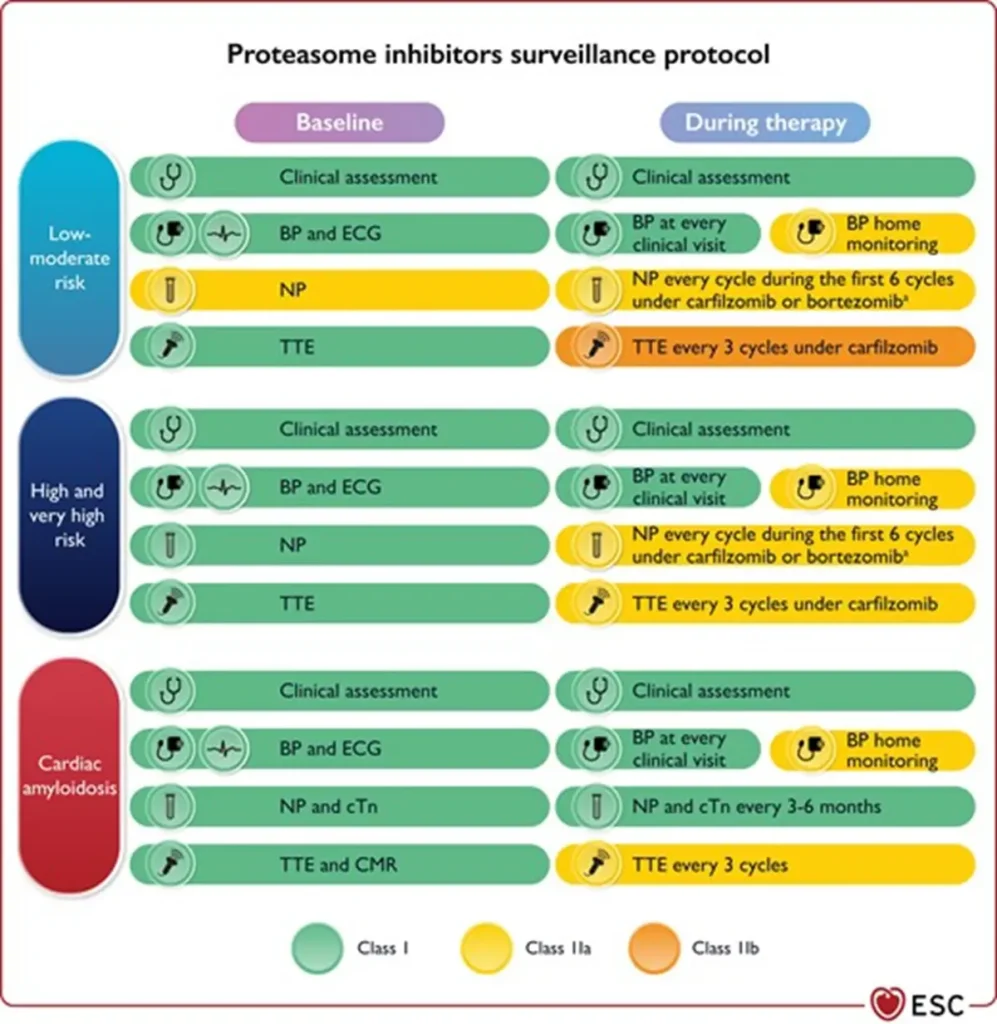
NTproBNP is not reimbursed in Belgium
Proposed Surveillance during multiple myeloma therapie (ESC guidelines):
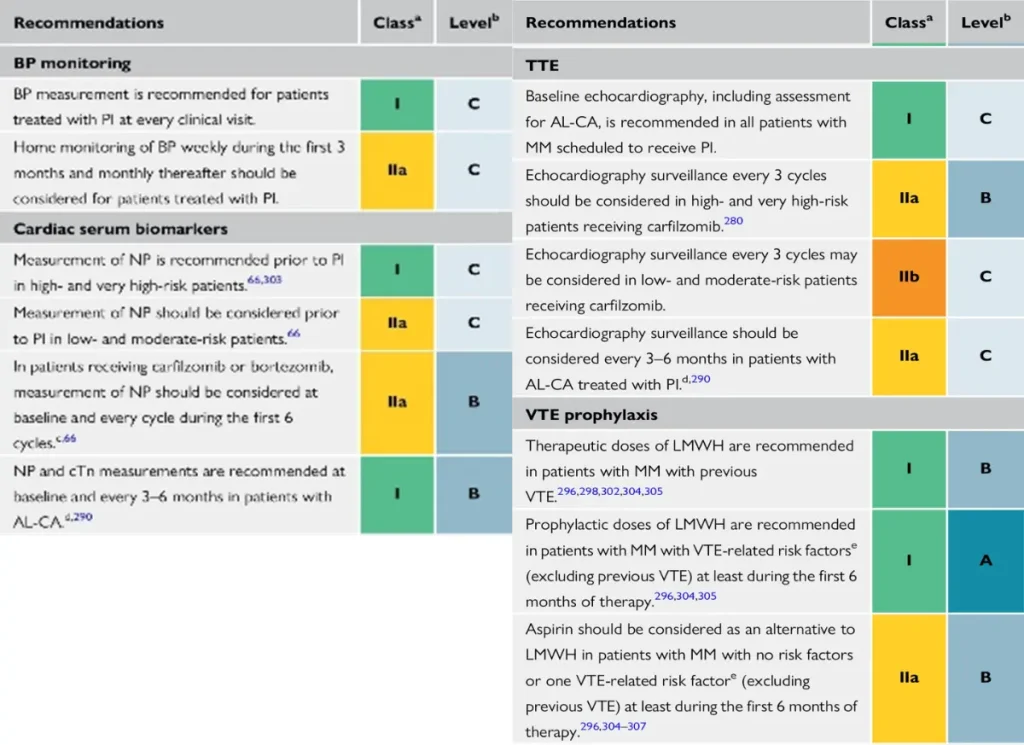
NTproBNP is not reimbursed in Belgium
References
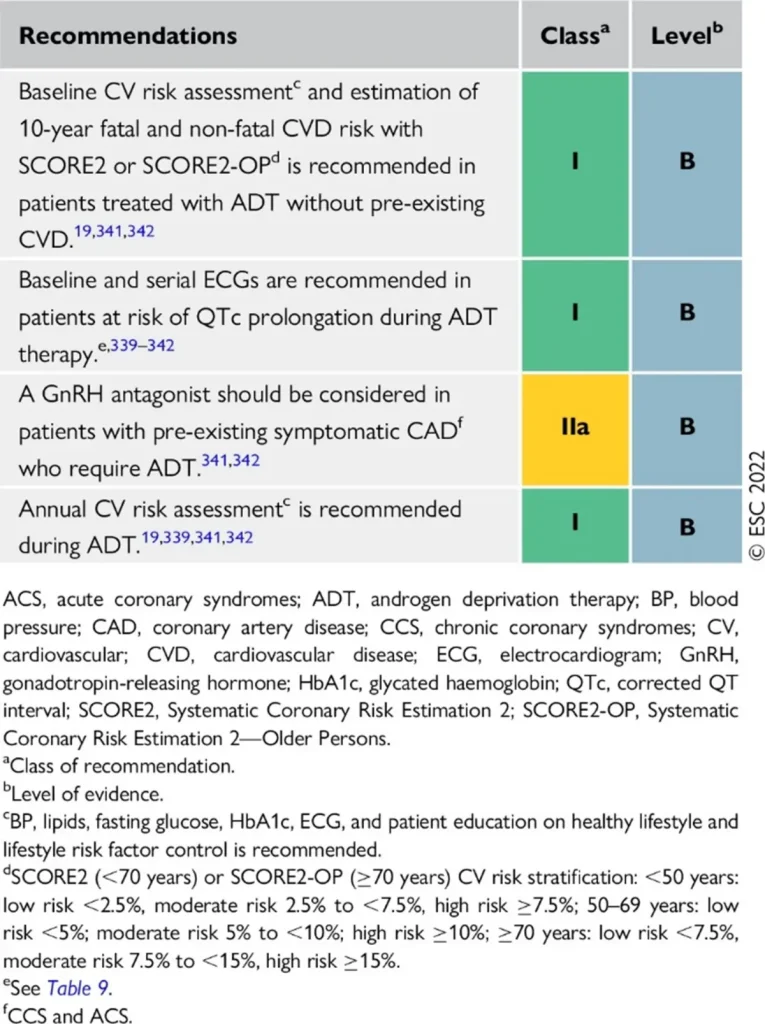
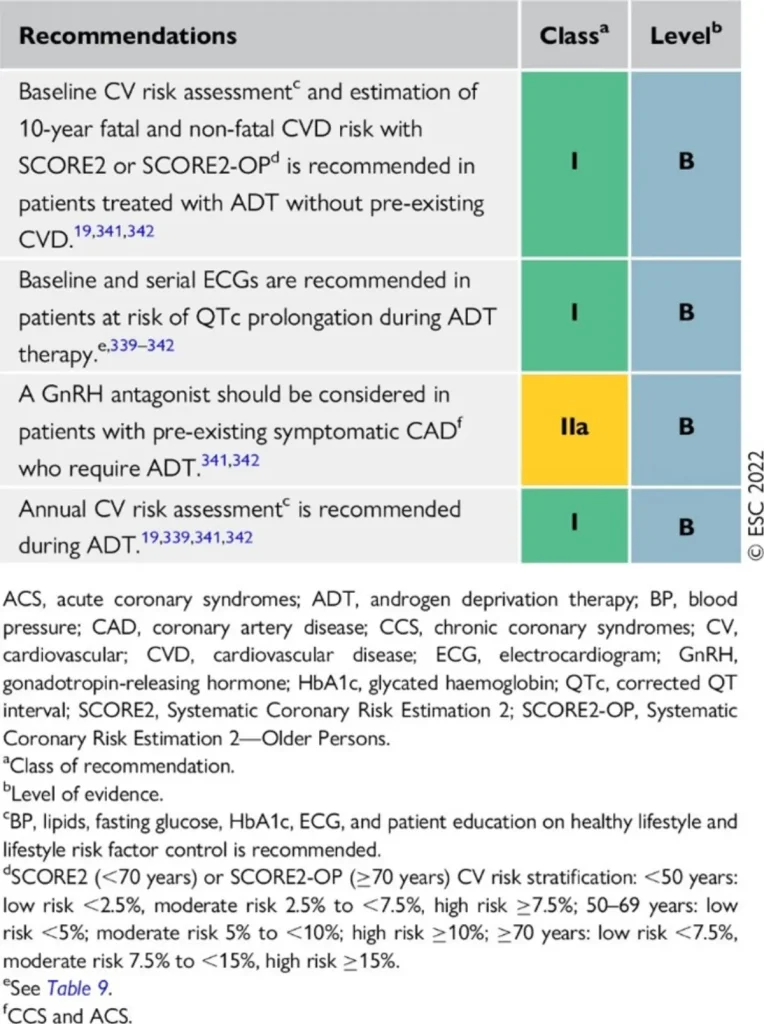
| Drug class / agents (commercial names) | Main oncologic indications | Type of cardiotoxicity | Approximate risk & surveillance |
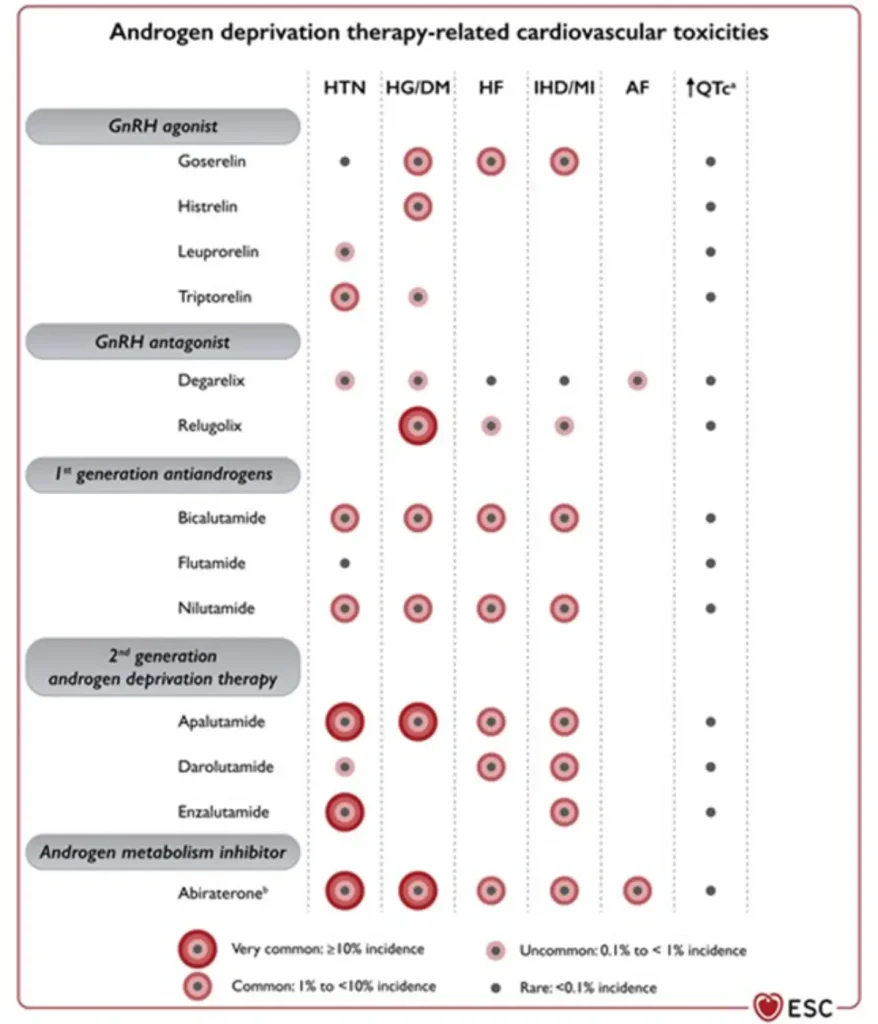
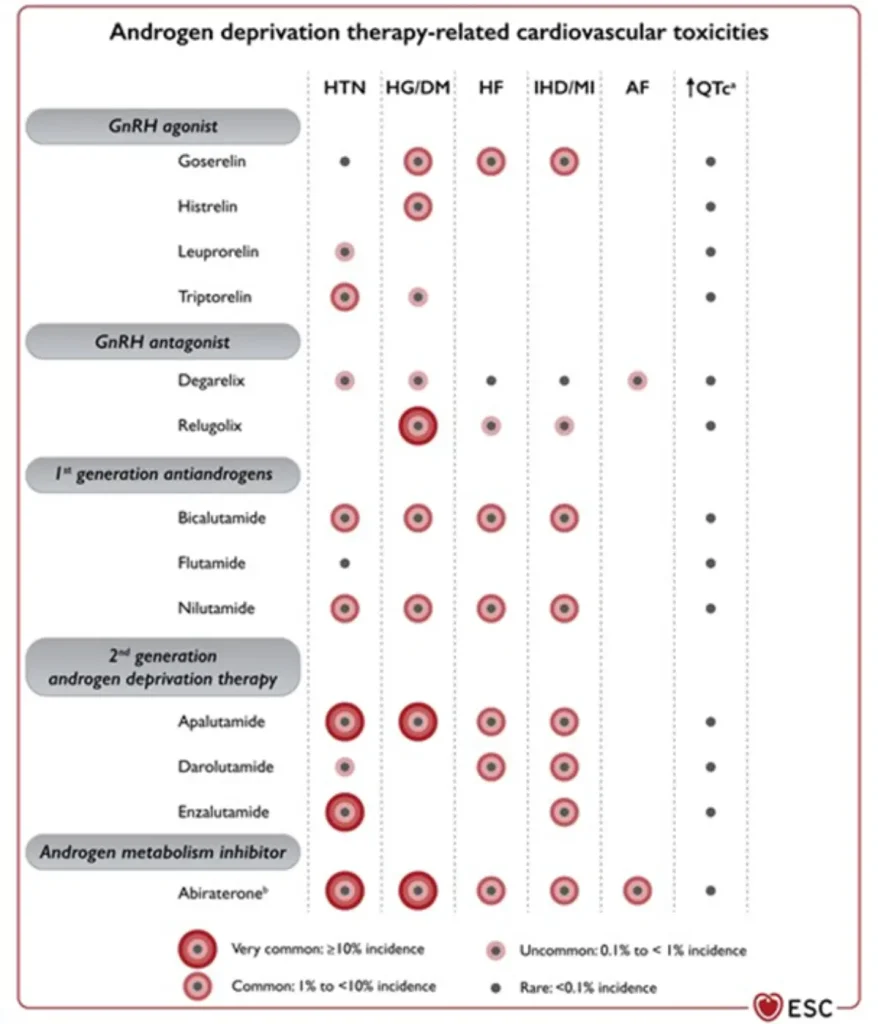
| GnRH agonists Leuprolide (Lucrin®, Eligard®) Goserelin (Zoladex®) Triptorelin (Decapeptyl®, Diphereline®) | Prostate cancer:
Breast cancer:
|
hypertension, hyperlipidaemia, hyperglycaemia / insulin resistance, metabolic syndrome
• Slight QTc prolongation | Cardiovascular events ~6–20% (higher in patients with pre-existing CV disease) |
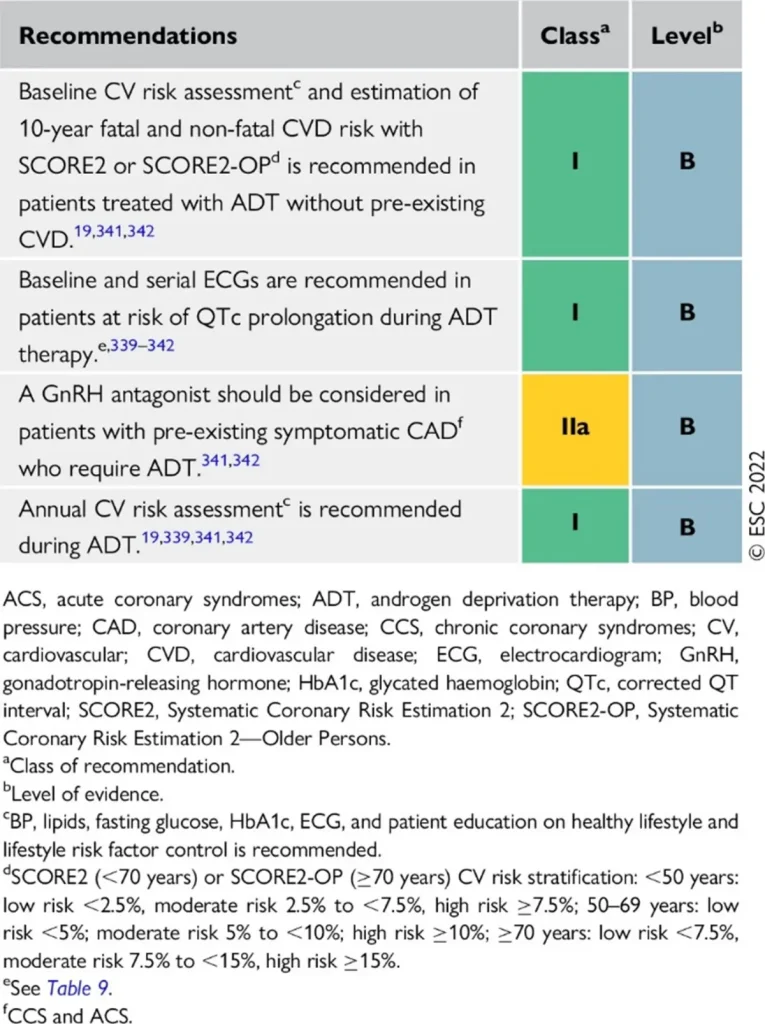
Proposed Surveillance (ESC guidelines):
References
| Drug class / agents (commercial names) | Main oncologic indications | ESC-aligned cardiotoxicity profile | Approximate risk & surveillance |
| GnRH antagonists Degarelix (Firmagon®) Relugolix (Orgovyx®) | Prostate cancer requiring rapid testosterone suppression, particularly in advanced or metastatic disease. |
| Cardiovascular events: ~3% Generally lower than with GnRH agonists |
GnRH antagonists are associated with a lower overall cardiovascular event rate compared with GnRH agonists, making them a preferred option in patients with high baseline cardiovascular risk, while still requiring structured cardiometabolic surveillance.
Proposed Surveillance (ESC guidelines):
References
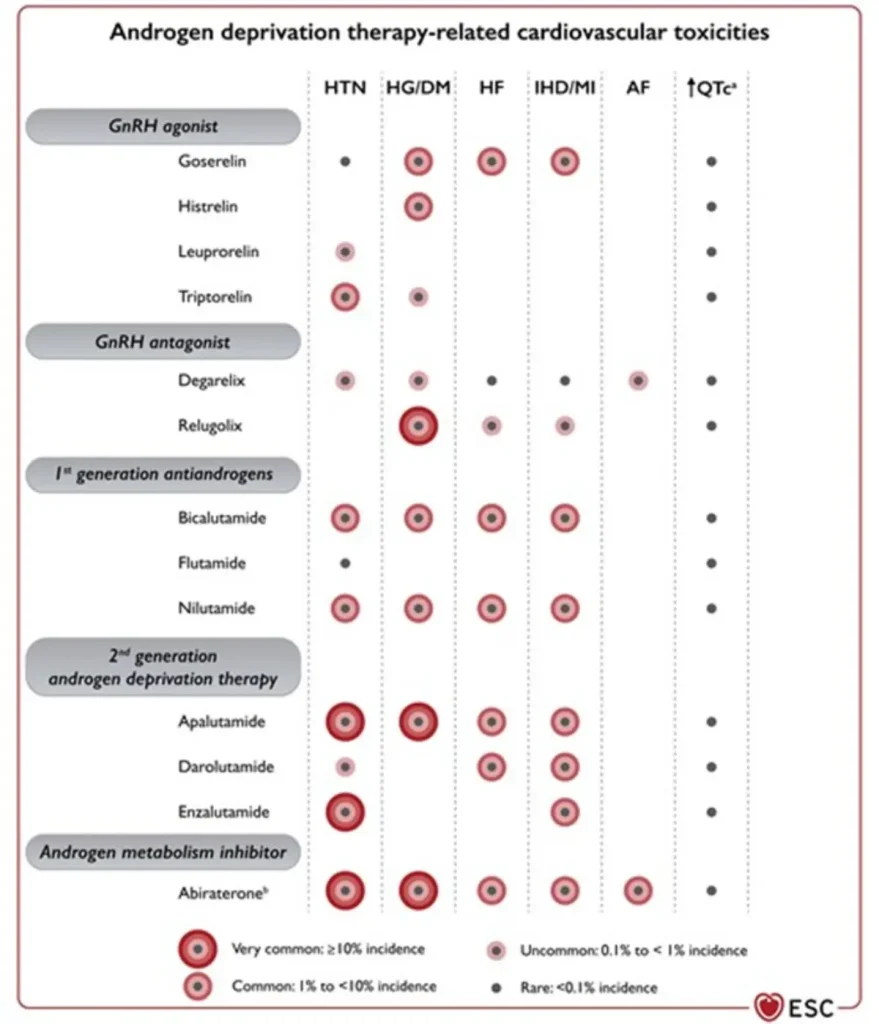
| Drug class / agents (commercial names) | Main oncologic indications | Type of cardiotoxicity | Approximate risk |
| ARTA – early generation (non-steroidal anti-androgens) Bicalutamide (Casodex®) Flutamide (Eulexin®) Nilutamide (Anandron®) – only rearely in use |
|
||
| ARTA – second generation Enzalutamide (Xtandi®) Apalutamide (Erleada®) Darolutamide (Nubeqa®) | Prostate cancer:
|
|
|
| Testosterone synthesis inhibitors Abiraterone acetate (Zytiga®) | Prostate cancer: • mHSPC • mCRPC (with prednisone) |
|
|
Androgen receptor–targeted agents substantially increase cardiovascular risk when added to ADT, with both any-grade and severe CV events occurring more frequently, underscoring the importance of baseline cardiovascular risk assessment, blood pressure control, and metabolic monitoring.
Proposed Surveillance (ESC guidelines):
References
| Drug class / agents | Main oncologic indications | Type of cardiotoxicity | Approximate risk |
BRAF inhibitors (BRAFi) Vemurafenib (Zelboraf®) MEK inhibitors (MEKi) Trametinib (Mekinist®) |
|
|
|
RAF/MEK inhibitor related cardiovascular toxicity
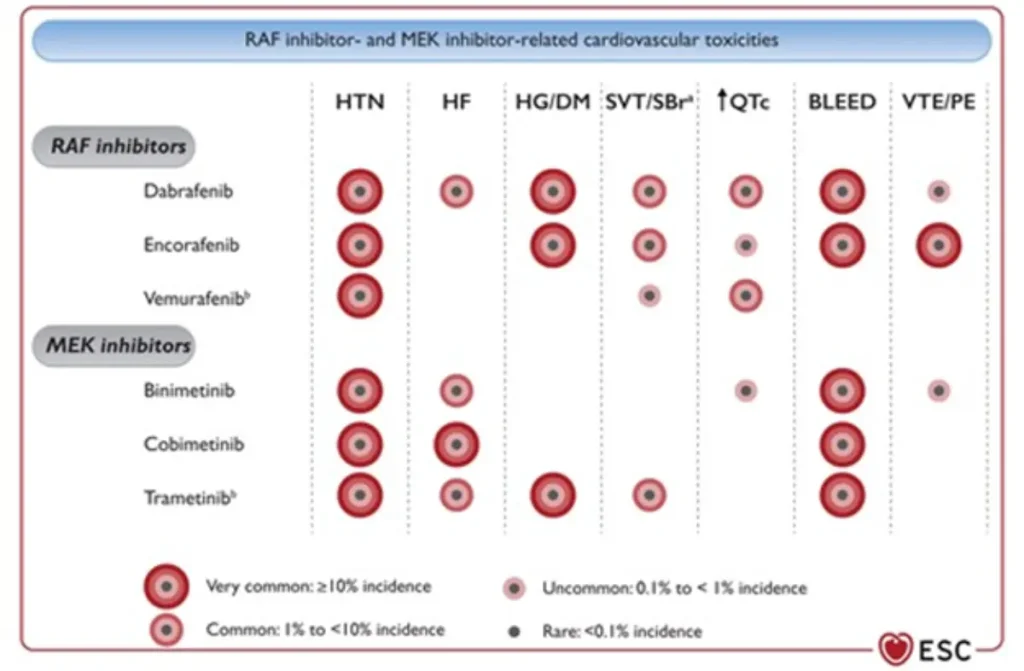
Proposed Surveillance (ESC guidelines):

Consider additional TTE 4 weeks after starting the therapy in high and very high risk patients, as most CTRCD with RAF/MEKi develop early.
References
| Drug class / agents | Main oncologic indications | Type of cardiotoxicity | Approximate risk (agent-specific where relevant) |
| CDK4/6 inhibitors Ribociclib (Kisqali®), Palbociclib (Ibrance®), Abemaciclib (Verzenios®) | Hormone receptor–positive, HER2-negative locally advanced or metastatic breast cancer, in combination with endocrine therapy. |
|
|
| PARP inhibitors (PARPi) Olaparib, Talazoparib | Germline BRCA1/2–mutated, HER2-negative breast cancer:
|
No consistent association with CTRCD, arrhythmias, or hypertension | |
In oncology patients, the Fridericia formula (QTcF = QT/RR¹ᐟ³) is preferred for QT correction because it reduces the overestimation of QT prolongation seen with Bazett’s formula — particularly at higher heart rates — and minimizes inappropriate management changes during QT-prolonging cancer therapy.
Proposed Surveillance for patients receiving CDK4/6i (ESC guidelines):
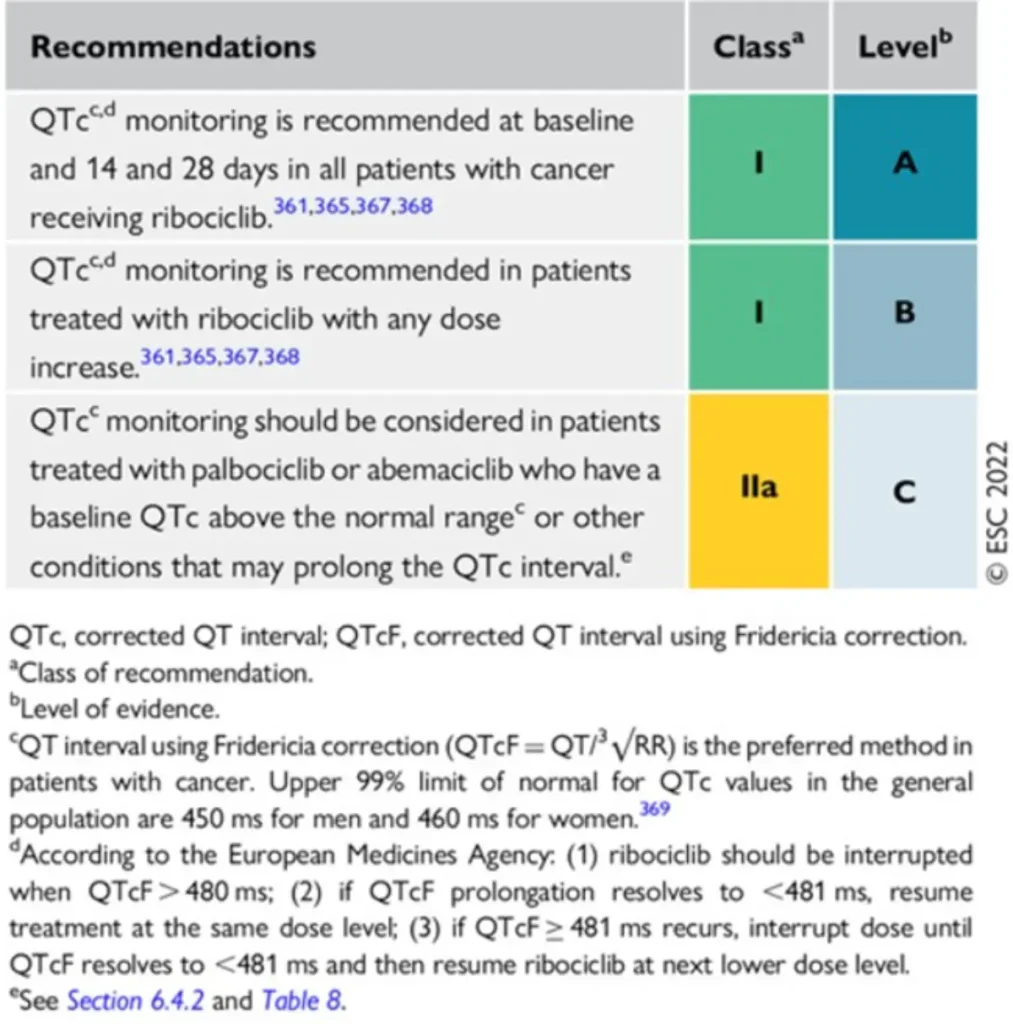
No cardiovascular surveillance is required for patients receiving PARPi
References
| Drug class / agents | Main oncologic / hematologic indications | Type of cardiotoxicity | Approximate risk (agent-specific where relevant) |
| BCR-ABL inhibitors Imatinib (Glivec®) Dasatinib (Sprycel®) Nilotinib (Tasigna®) Ponatinib (Iclusig®) Bosutinib (Bosulif®) | Hematologic malignancies:
|
|
LV dysfunction / CHF:
|
BCR-ABL inhibitors related cardiovascular toxicities
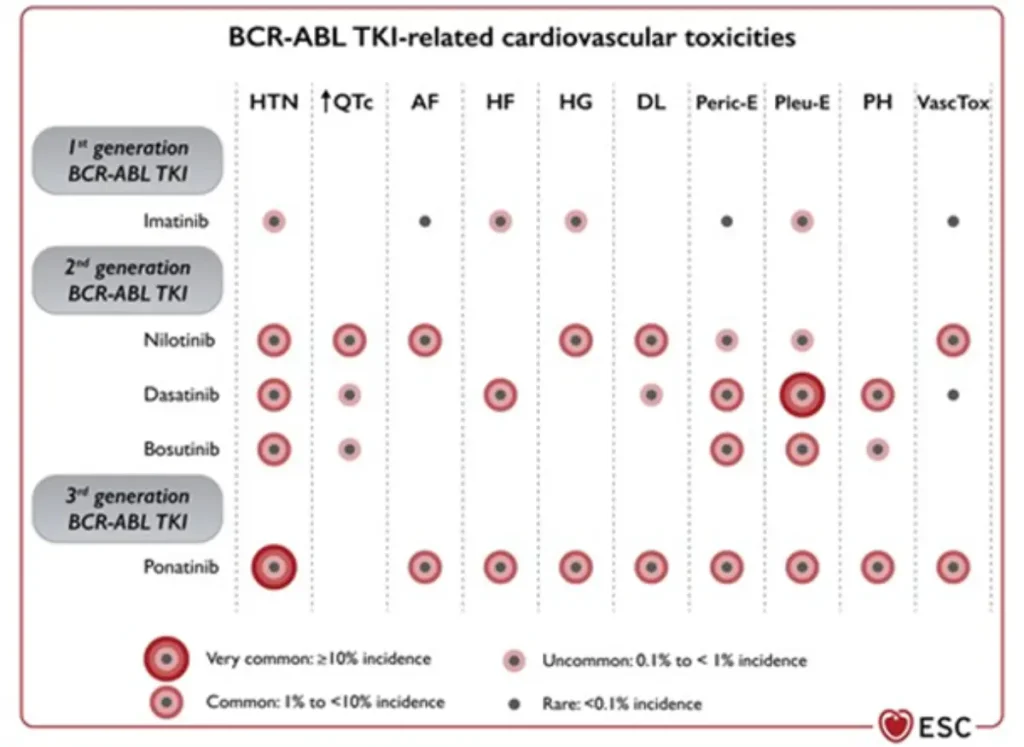
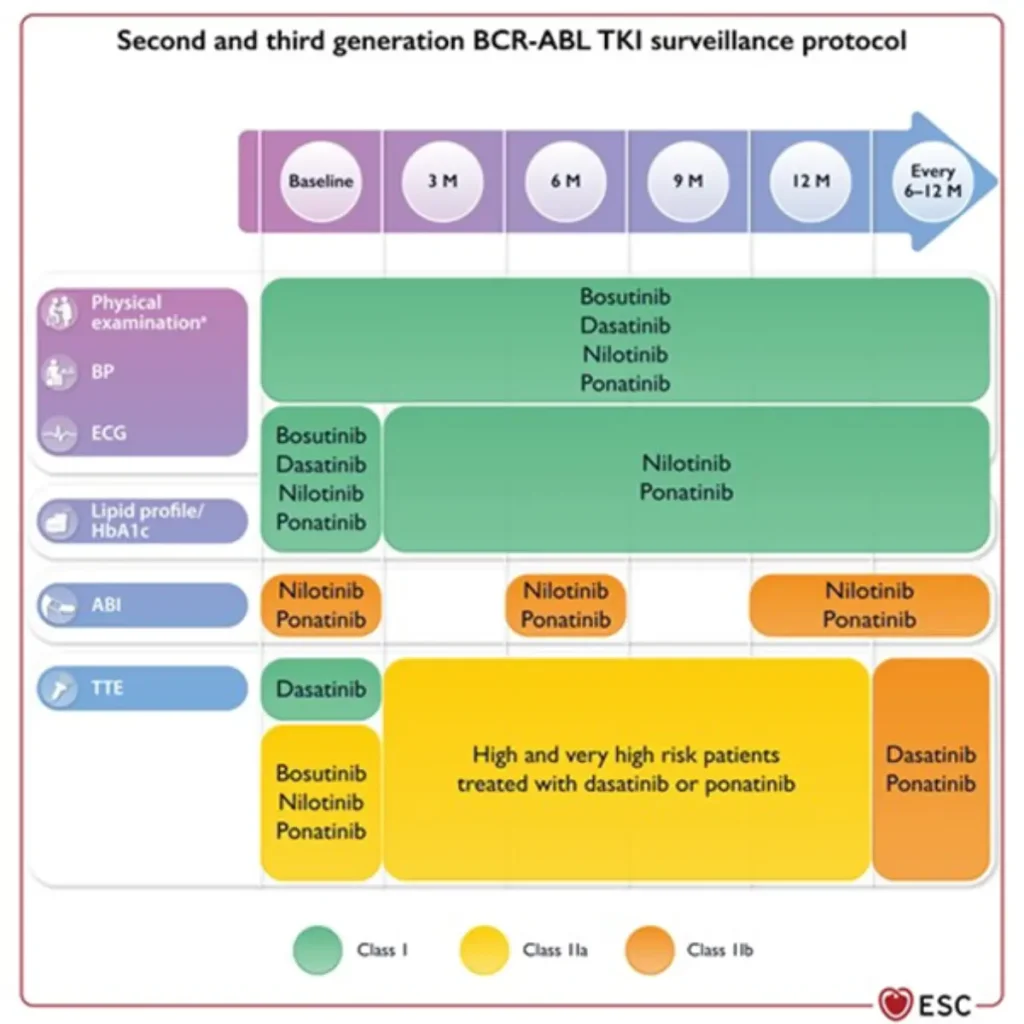
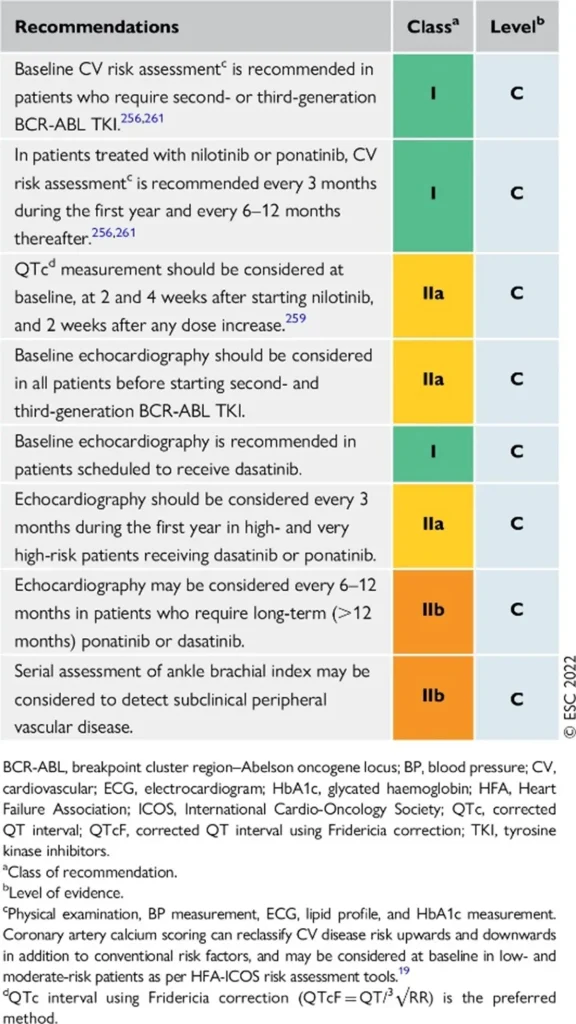
Proposed Surveillance (ESC guidelines):
References
| Drug class / agents | Main oncologic indications | Type of cardiotoxicity | Approximate risk |
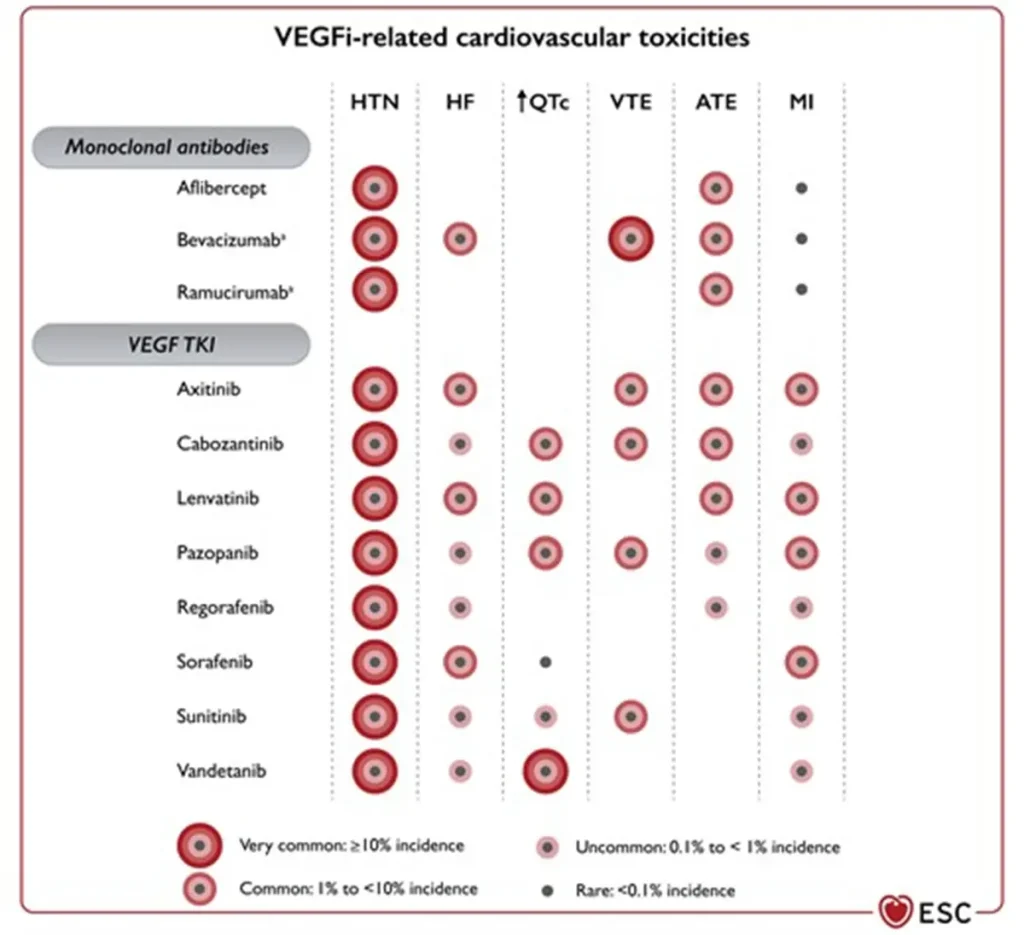
Vascular endothelial growth factor inhibitors VEGF (VEGFi) Sunitinib (Sutent®) Sorafenib (Nexavar®) Pazopanib (Votrient®) Axitinib (Inlyta®) Lenvatinib (Lenvima®) Cabozantinib (Cabometyx® / Cometriq®) Regorafenib (Stivarga®) Vandetanib (Caprelsa®) |
|
|
|
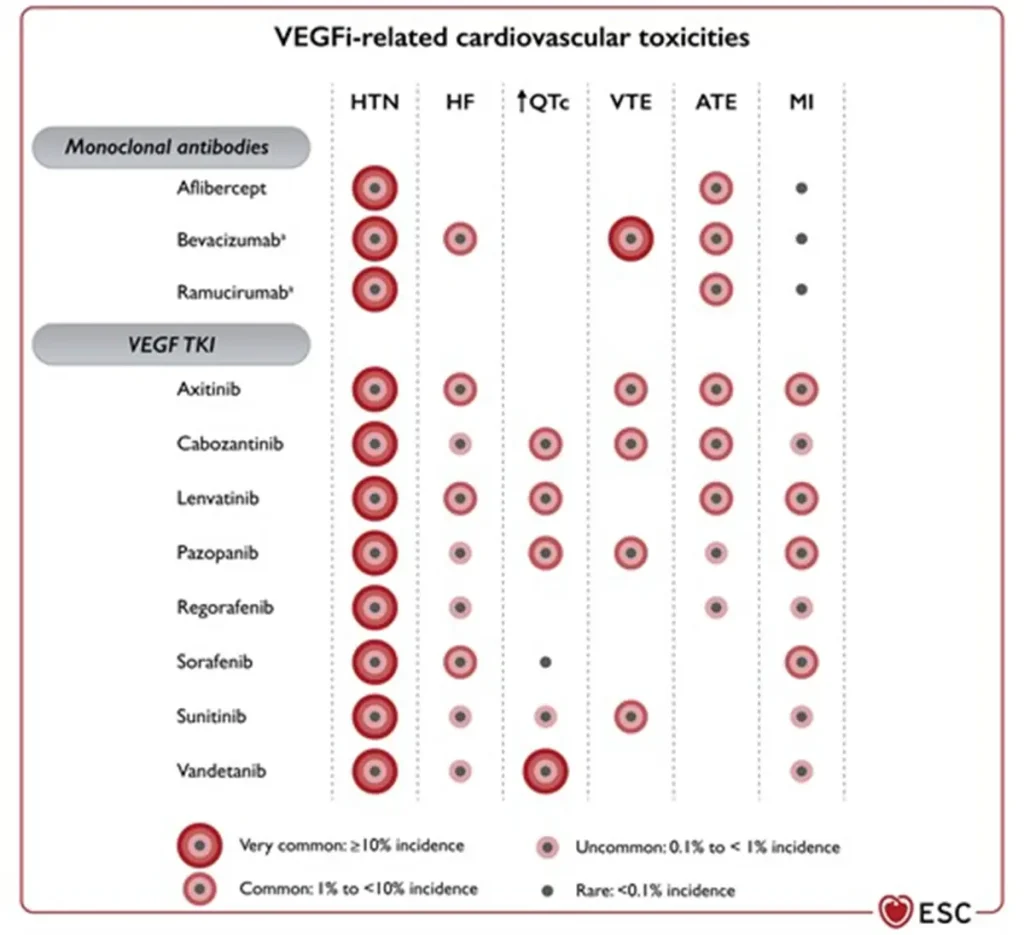
Angiogenesis inhibitor related cardiovascular toxicities
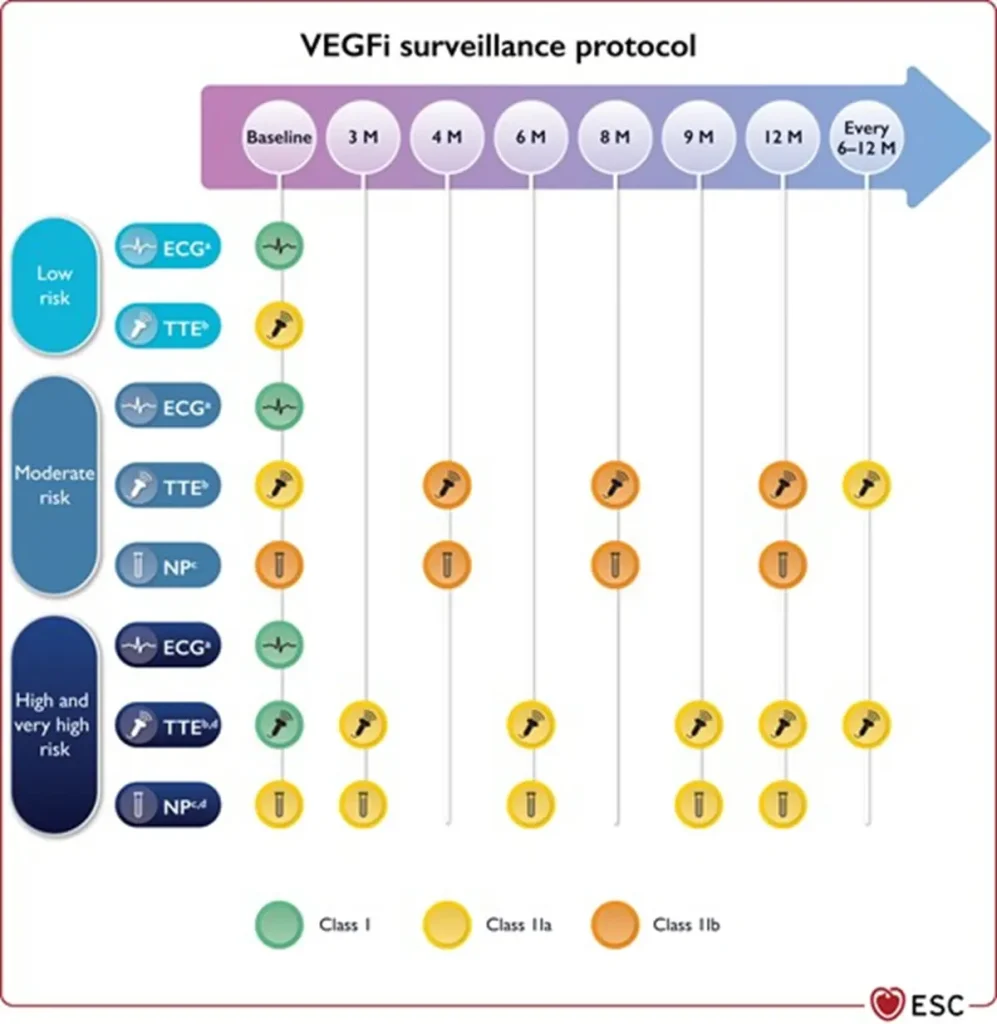
Proposed Surveillance (ESC guidelines):
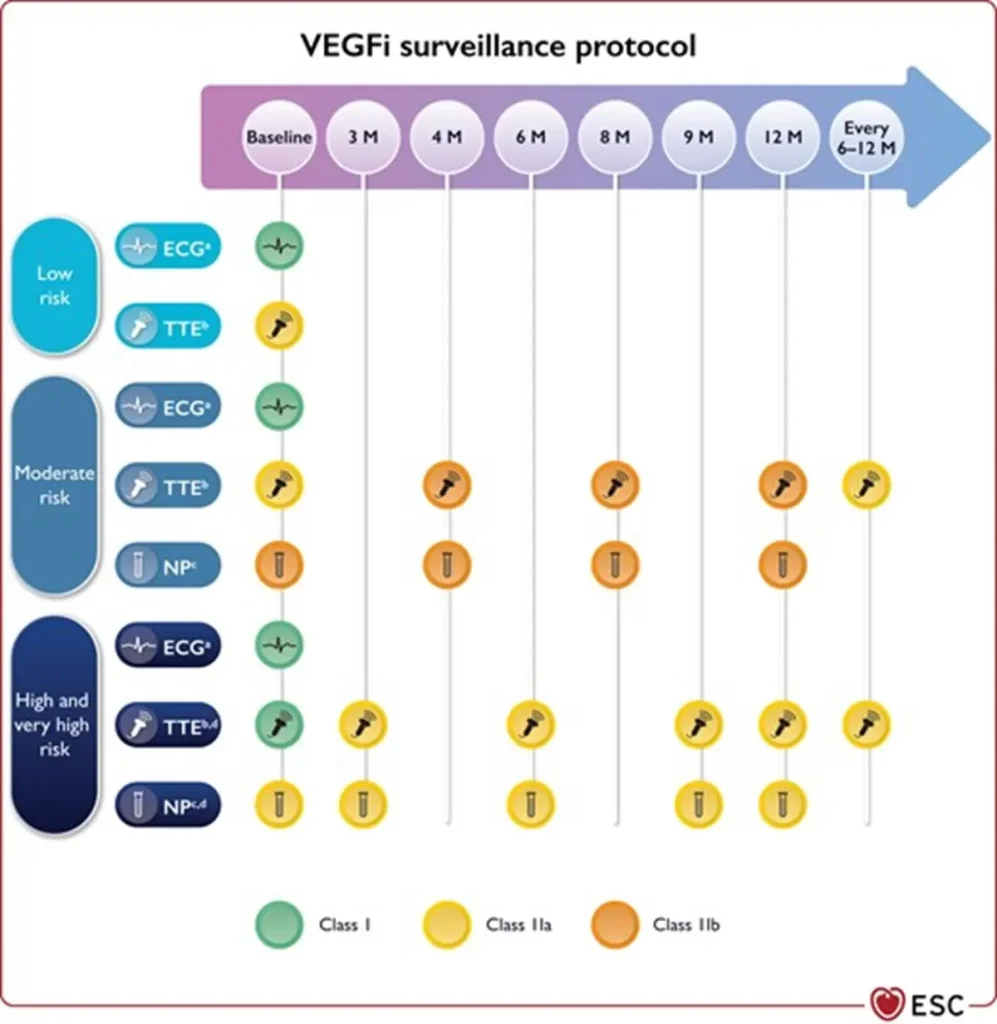
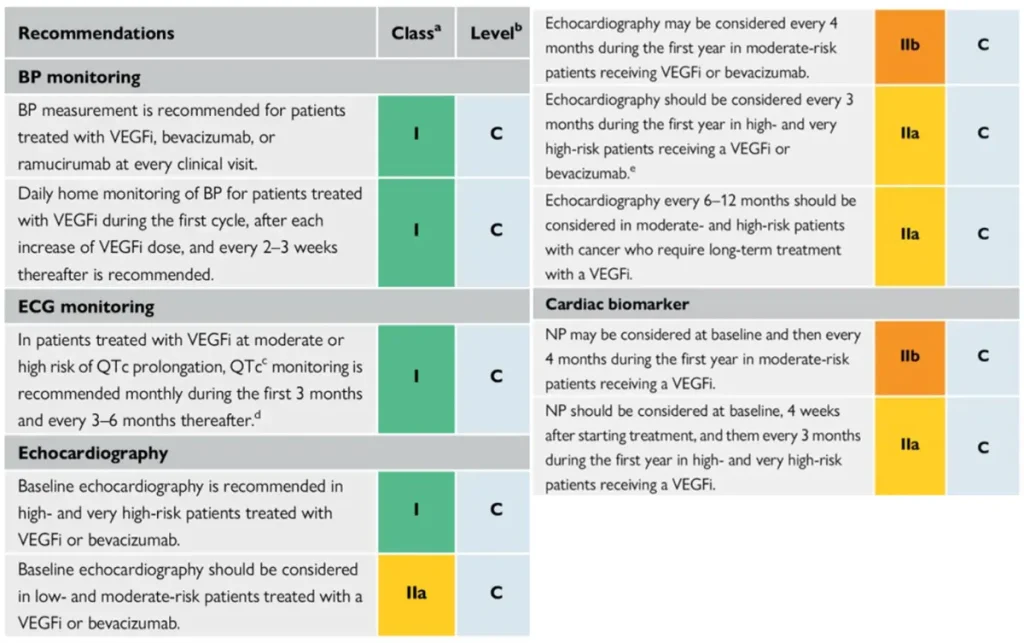
NTproBNP is not reimbursed in Belgium
References
| Drug class / agents | Main oncologic indications | Type of cardiotoxicity | Approximate risk & surveillance |
| ALK inhibitors Crizotinib, Ceritinib, Alectinib, Brigatinib, Lorlatinib | ALK-positive NSCLC. ROS1-positive NSCLC (selected agents). Other rare ALK-driven tumours. |
| Bradycardia: ~10–20%, often asymptomatic. QT prolongation: ~5–10%. |
| EGFR inhibitors Erlotinib, Lapatinib | EGFR-mutated NSCLC (erlotinib). HER2-positive metastatic breast cancer (lapatinib, usually with capecitabine). |
| Overall low cardiovascular risk (<2–3%) |
Surveillance :
ALK and EGFR inhibitors are non-angiogenic TKIs with a cardiovascular toxicity profile dominated by electrical disturbances rather than vascular toxicity, and generally require limited routine cardiac surveillance. Individualized approach is therefore recommended
References
| Drug class / agents | Main hematologic indications | Type of cardiotoxicity | Approximate risk (agent-specific where relevant) |
| BTK inhibitors Ibrutinib (Imbruvica®) Acalabrutinib (Calquence®) Zanubrutinib (Brukinsa®) | B-cell malignancies:
|
|
|
Bleeding is very common, particularly with ibrutinib (up to ~55%), due to off-target antiplatelet effects. Individualized risk–benefit assessment is required in patients needing antiplatelet or anticoagulant therapy, especially dual antiplatelet therapy.
Proposed Surveillance (ESC guidelines) :
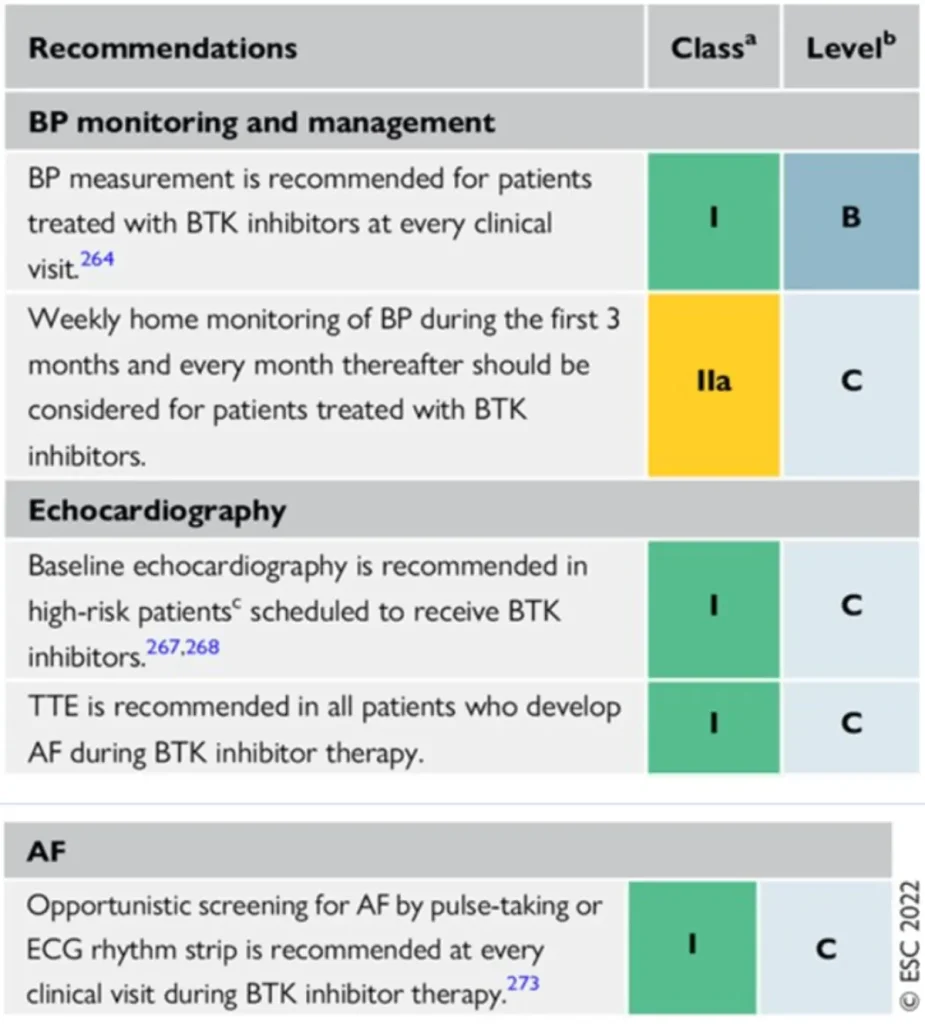
References
| Drug class / agents | Main oncologic indications | Type of cardiotoxicity | Approximate risk |
| anti-VEGF monoclonal antibodies Bevacizumab (Avastin®) |
|
|
|
Vascular endothelial growth factor inhibitors VEGF (VEGFi) Sunitinib (Sutent®) Sorafenib (Nexavar®) Pazopanib (Votrient®) Axitinib (Inlyta®) Lenvatinib (Lenvima®) Cabozantinib (Cabometyx® / Cometriq®) Regorafenib (Stivarga®) Vandetanib (Caprelsa®) |
|
|
|
Angiogenesis inhibitor related cardiovascular toxicities
Proposed Surveillance (ESC guidelines) :
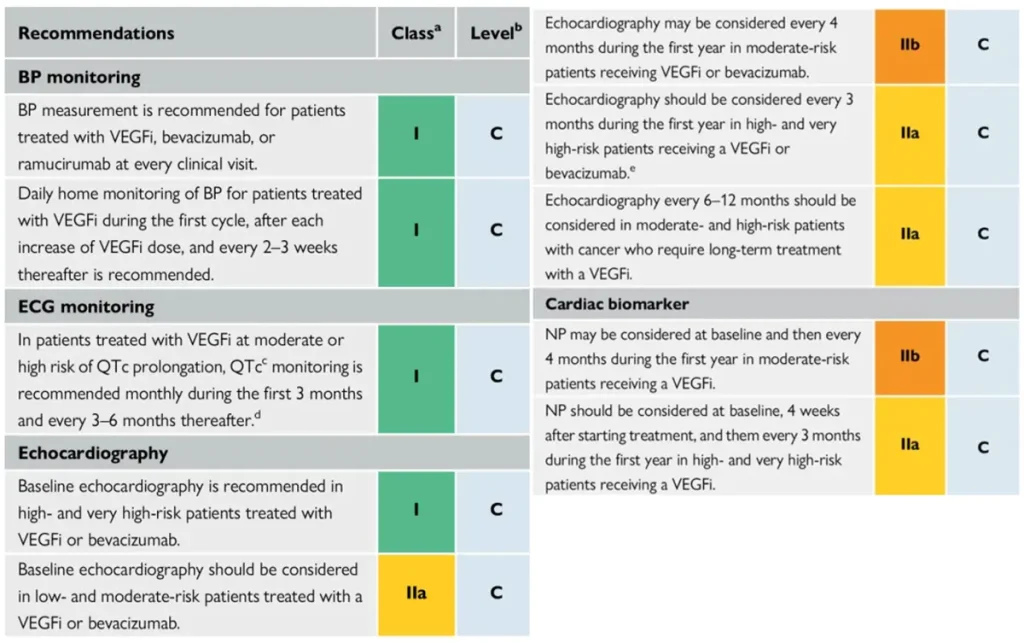
NTproBNP is not reimbursed in Belgium
References
| Drug class / agents (commercial names) | Main oncologic indications | Type of cardiotoxicity | Approximate risk |
| Aromatase inhibitors (AIs) Anastrozole (Arimidex®) Letrozole (Femara®) Exemestane (Aromasin®) |
|
|
|
Selective estrogen receptor modulators (SERMs) Tamoxifen (Nolvadex®) Raloxifene (Evista®) |
|
|
|
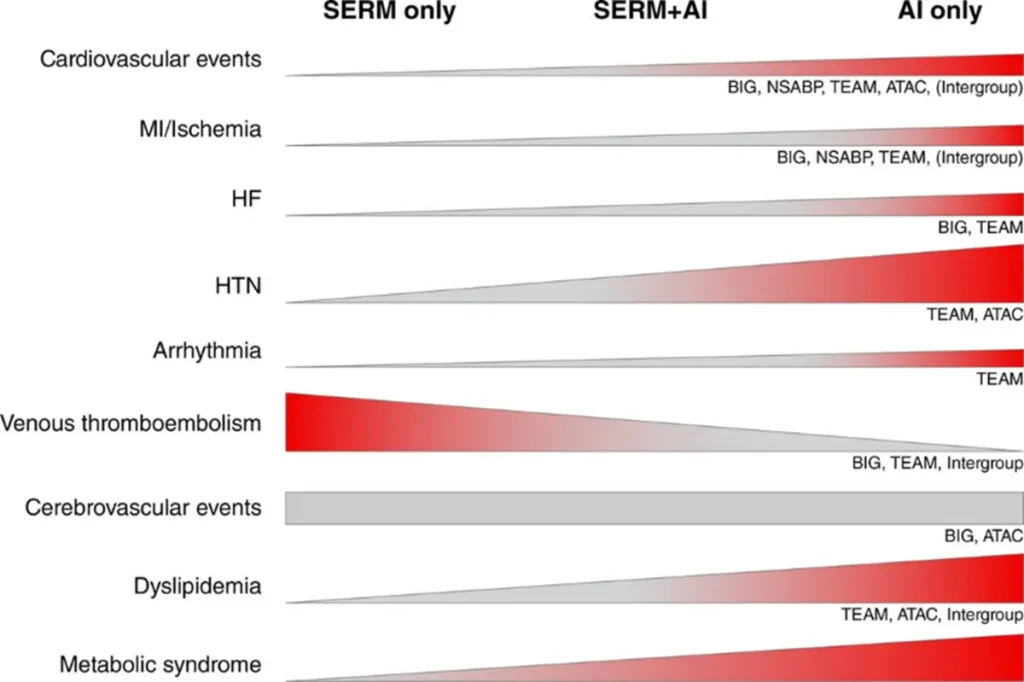
Okwuosa TM et al. June 2021
Proposed Surveillance (ESC guidelines) :
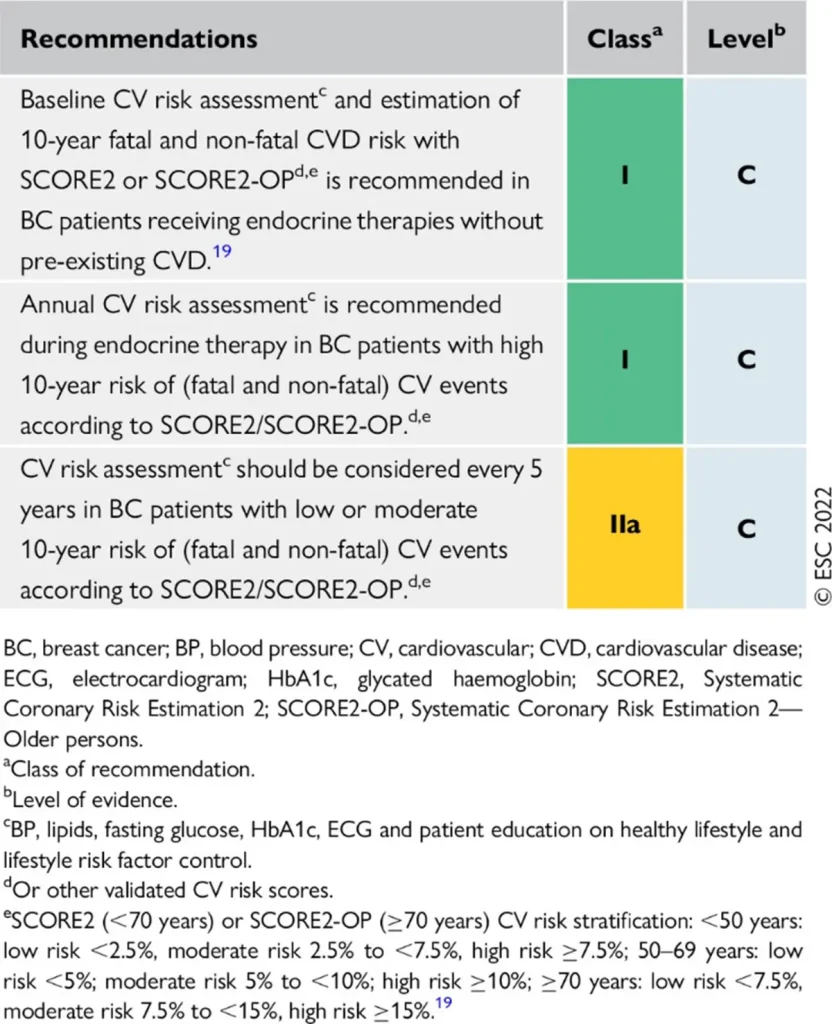
References